On April 1, 2026, the U.S. Food and Drug Administration officially approved Eli Lilly’s novel oral weight-loss drug, Founday (orforglipron). Orforglipron has become the world’s first GLP-1 receptor agonist that can be taken “anytime, anywhere, without any restrictions on food or water intake.”
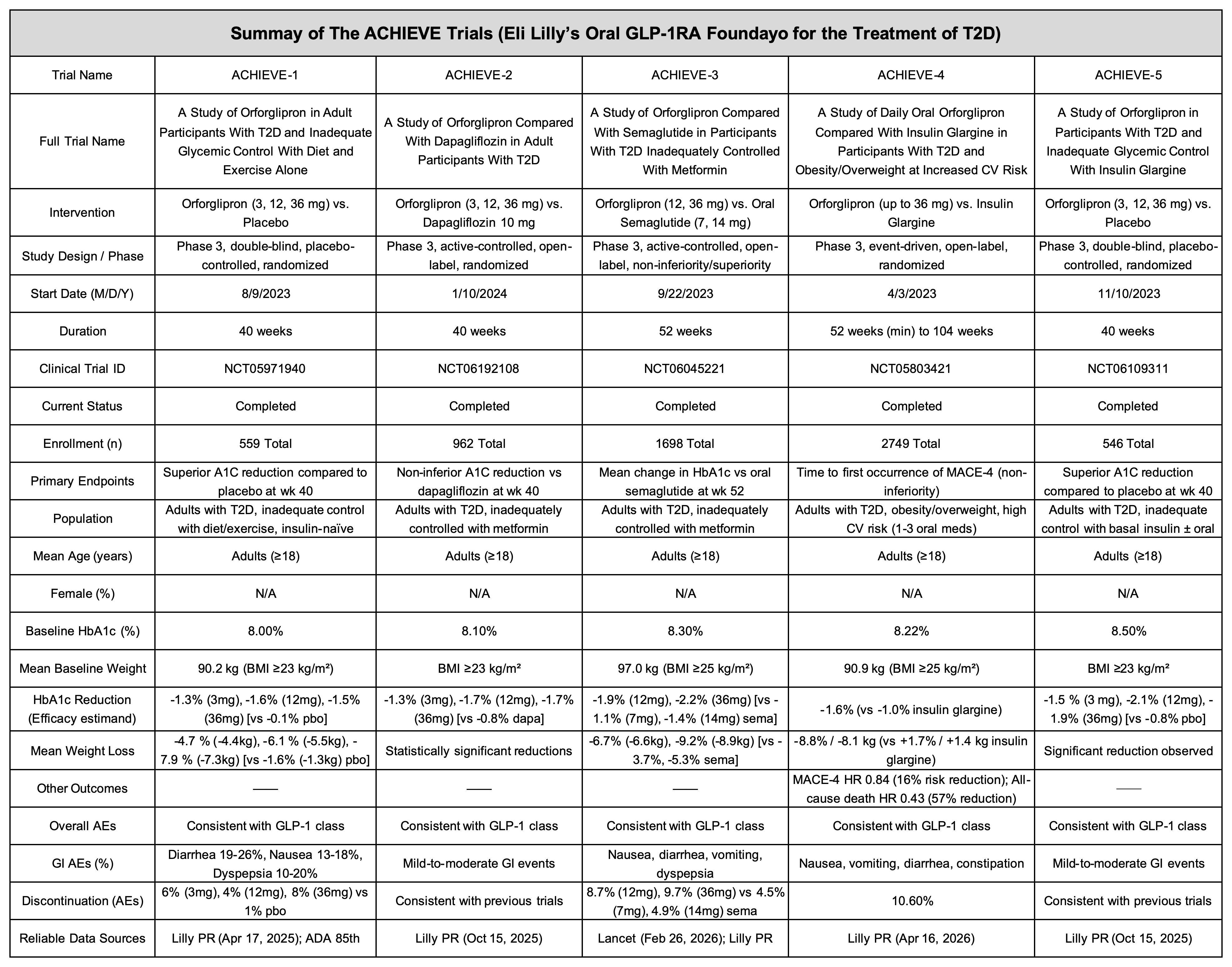
At the same time, to fully benchmark against semaglutide, Eli Lilly has constructed an extensive and rigorous clinical development program for Orforglipron in type 2 diabetes (T2D): the global Phase 3 ACHIEVE program. This program includes multiple large-scale head-to-head trials comparing Orforglipron with current standard-of-care therapies, enrolling over 6,000 T2D patients in total. This article summarizes the key clinical trials within the ACHIEVE program, including timelines, study designs, clinical data, and data sources.
ACHIEVE-1 Study
The natural course of T2D is progressive. In early stages, patients may maintain glycemic control through diet and exercise, but as pancreatic β-cell function declines, lifestyle interventions eventually fail.
ACHIEVE-1 (NCT05971940) specifically targets this population: early-stage T2D patients who have never received insulin and can no longer achieve adequate glycemic control through lifestyle modification alone. These patients had relatively healthy baseline characteristics, with a mean HbA1c of 8.0% and an average body weight of 90.2 kg.
In this 40-week, randomized, double-blind, placebo-controlled trial, 559 patients were enrolled. Using the efficacy estimand model:
- HbA1c reductions:
- 3 mg: −1.3%
- 12 mg: −1.6%
- 36 mg: −1.5%
- Placebo: −0.1%
- 3 mg: −1.3%
- 12 mg: −1.6%
- 36 mg: −1.5%
- Placebo: −0.1%
Notably, over 65% of patients in the 36 mg group achieved HbA1c ≤6.5%. According to the American Diabetes Association, 6.5% is the diagnostic threshold for diabetes, meaning more than 60% of patients returned to non-diabetic glycemic levels with a non-injectable therapy.
Weight reduction:
- 3 mg: −4.4 kg (4.7%)
- 12 mg: −5.5 kg (6.1%)
- 36 mg: −7.3 kg (7.9%)
- Placebo: −1.3 kg
Safety data confirmed feasibility for chronic use. Gastrointestinal side effects were observed (diarrhea 26%, nausea 16%), but discontinuation rates due to adverse events were ≤8%, even at the highest dose.
ACHIEVE-1 clearly demonstrates that once-daily oral therapy can match or even surpass traditional first-line injectable treatments in early T2D.
Head-to-Head Superiority: ACHIEVE-2 and ACHIEVE-3
ACHIEVE-2: Outperforming SGLT2 Inhibitor Dapagliflozin
SGLT2 inhibitors such as Dapagliflozin lower glucose by promoting urinary excretion and are widely used as second-line therapies.
ACHIEVE-2 (NCT06192108) enrolled 962 patients inadequately controlled on metformin, comparing Orforglipron with 10 mg dapagliflozin.
Although designed for non-inferiority, the results showed clear superiority:
- Baseline HbA1c: 8.1%
- HbA1c reduction:
- Dapagliflozin: −0.8%
- Orforglipron:
- 3 mg: −1.3%
- 12 mg: −1.7%
- 36 mg: −1.7%
- Dapagliflozin: −0.8%
- Orforglipron:
- 3 mg: −1.3%
- 12 mg: −1.7%
- 36 mg: −1.7%
- 3 mg: −1.3%
- 12 mg: −1.7%
- 36 mg: −1.7%
This nearly twofold difference highlights the stronger systemic hormonal regulation of GLP-1 versus renal glucose excretion mechanisms.
ACHIEVE-3: The Ultimate Oral GLP-1 Showdown
ACHIEVE-3 (NCT06045221) compared Orforglipron with oral semaglutide in a 52-week global study involving 1,698 patients—the first direct comparison of oral GLP-1 therapies.
Unlike semaglutide, which requires strict fasting conditions, Orforglipron has no intake restrictions.
Results:
- HbA1c reduction:
- Orforglipron 36 mg: −2.2%
- Oral semaglutide 14 mg: −1.4%
- Orforglipron 36 mg: −2.2%
- Oral semaglutide 14 mg: −1.4%
- Weight loss:
- Orforglipron: −19.7 lbs (9.2%)
- Semaglutide: −11.0 lbs (5.3%)
- Orforglipron: −19.7 lbs (9.2%)
- Semaglutide: −11.0 lbs (5.3%)
This represents a 73.6% greater weight reduction with Orforglipron.
These findings, published in The Lancet, attracted significant attention. The combination of superior efficacy, dosing flexibility, and improved adherence suggests that small-molecule GLP-1 therapies may disrupt the peptide-based SNAC technology paradigm.
Advanced Disease & Outcomes: ACHIEVE-4 and ACHIEVE-5
ACHIEVE-4: Cardiovascular and Survival Benefits
ACHIEVE-4 (NCT05803421) is a large-scale (>2,700 patients), event-driven cardiovascular outcomes trial (CVOT) in T2D patients with obesity/overweight and high cardiovascular risk.
The most striking result:
- All-cause mortality reduced by 57% vs insulin glargine
(HR 0.43, p=0.002)
This survival benefit may stem from GLP-1–mediated improvements in endothelial function, plaque stability, and reductions in systemic inflammation (e.g., hsCRP) and non-HDL cholesterol.
ACHIEVE-4 demonstrates that Orforglipron is not merely a glucose-lowering drug, but a comprehensive metabolic regulator capable of reshaping cardiovascular health and extending lifespan.
ACHIEVE-5: Combination with Basal Insulin
ACHIEVE-5 (NCT06109311) evaluated Orforglipron added to insulin glargine in 546 patients with baseline HbA1c of 8.5%.
After 40 weeks:
- HbA1c reduction:
- 12 mg: −2.1%
- 36 mg: −1.9%
- Placebo: −0.8%
- 12 mg: −2.1%
- 36 mg: −1.9%
- Placebo: −0.8%
Importantly, Orforglipron offset insulin-induced weight gain, achieving meaningful weight loss while enhancing glycemic control.
This provides a powerful combination strategy: improved glucose control without increasing hypoglycemia risk or weight burden.
Conclusion
Across the ACHIEVE program, Orforglipron has demonstrated its potential as a next-generation cornerstone therapy for T2D.
As Eli Lilly prepares regulatory submissions to the FDA and global agencies in 2026, Orforglipron is poised to usher in a new era of diabetes treatment—defined by high efficacy, oral administration, and seamless patient adherence.