On March 28, at the 2026 American Academy of Dermatology (AAD) Annual Meeting, Biogen announced a major milestone: its investigational innovative therapy, Litifilimab (BIIB059), successfully met the primary endpoint in the Phase 2 portion (Part A) of the AMETHYST Phase II/III clinical study for Cutaneous Lupus Erythematosus (CLE), significantly reducing skin disease activity in patients.
This marks the second positive Phase 2 clinical result for Litifilimab. As the only investigational project to demonstrate consistent, positive efficacy across multiple CLE studies to date, Litifilimab is poised to potentially become the first targeted therapy for the disease in over 70 years.
About Litifilimab
Developed internally by Biogen, Litifilimab is a first-in-class humanized IgG1 monoclonal antibody targeting Blood Dendritic Cell Antigen 2 (BDCA2). BDCA2 is primarily expressed on the surface of plasmacytoid dendritic cells (pDCs). Research indicates that by binding to BDCA2, Litifilimab effectively inhibits the production of pro-inflammatory molecules, including Type I Interferons (IFN-I), which are key drivers in the pathogenesis of both systemic and cutaneous lupus erythematosus.
Due to its exceptional early-stage data, Litifilimab previously received Breakthrough Therapy Designation from the U.S. FDA.
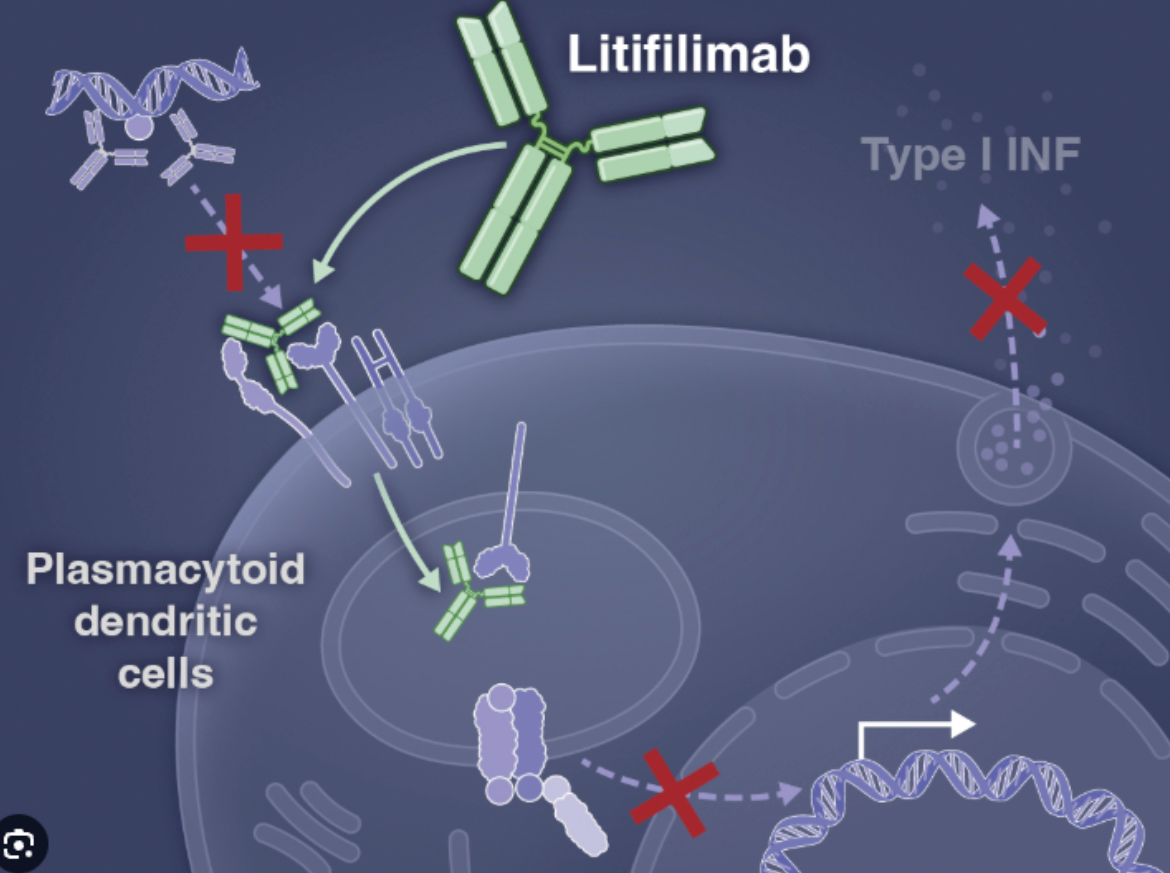
Clinical Data: Rapid Onset and Significant Lesion Improvement
The AMETHYST study is an ongoing, seamless two-stage, multicenter, double-blind, placebo-controlled randomized trial. The data from Part A (Weeks 0-24) are highly encouraging:
-
Primary Endpoint Achieved: At Week 16, as measured by the Cutaneous Lupus Erythematosus Investigator’s Global Assessment-Revised (CLA-IGA-R) Erythema Score (0-1, indicating skin lesions cleared or almost cleared), the proportion of patients with reduced disease activity in the Litifilimab group reached 14.7%, compared to just 2.9% in the placebo group (a difference of 11.8%, p < 0.05).
-
Rapid Onset and Sustained Efficacy: Separation from placebo was observed as early as Week 4. By Week 24, 40.8% of patients in the Litifilimab group achieved a CLASI-50 response (a 50% improvement from baseline in CLASI-A score), compared to 21% in the placebo group.
-
Higher Proportion of Deep Remission: At Week 24, 21.7% of patients in the Litifilimab group achieved a CLASI-70 response (vs. 5.8% for placebo). Furthermore, one-sixth (16.3%) of the Litifilimab group reached a CLASI score of 0-3 (representing no or minimal disease activity), while the placebo group remained at 0%.
-
Favorable Safety and Tolerability: In Part A of the AMETHYST study, Litifilimab demonstrated a favorable safety profile. Through 24 weeks, the incidence of adverse events (AEs) was 74.6% for the Litifilimab group and 64.7% for the placebo group, with the vast majority being mild to moderate. Serious adverse events (SAEs) occurred at rates of 6.8% and 2.9%, respectively.
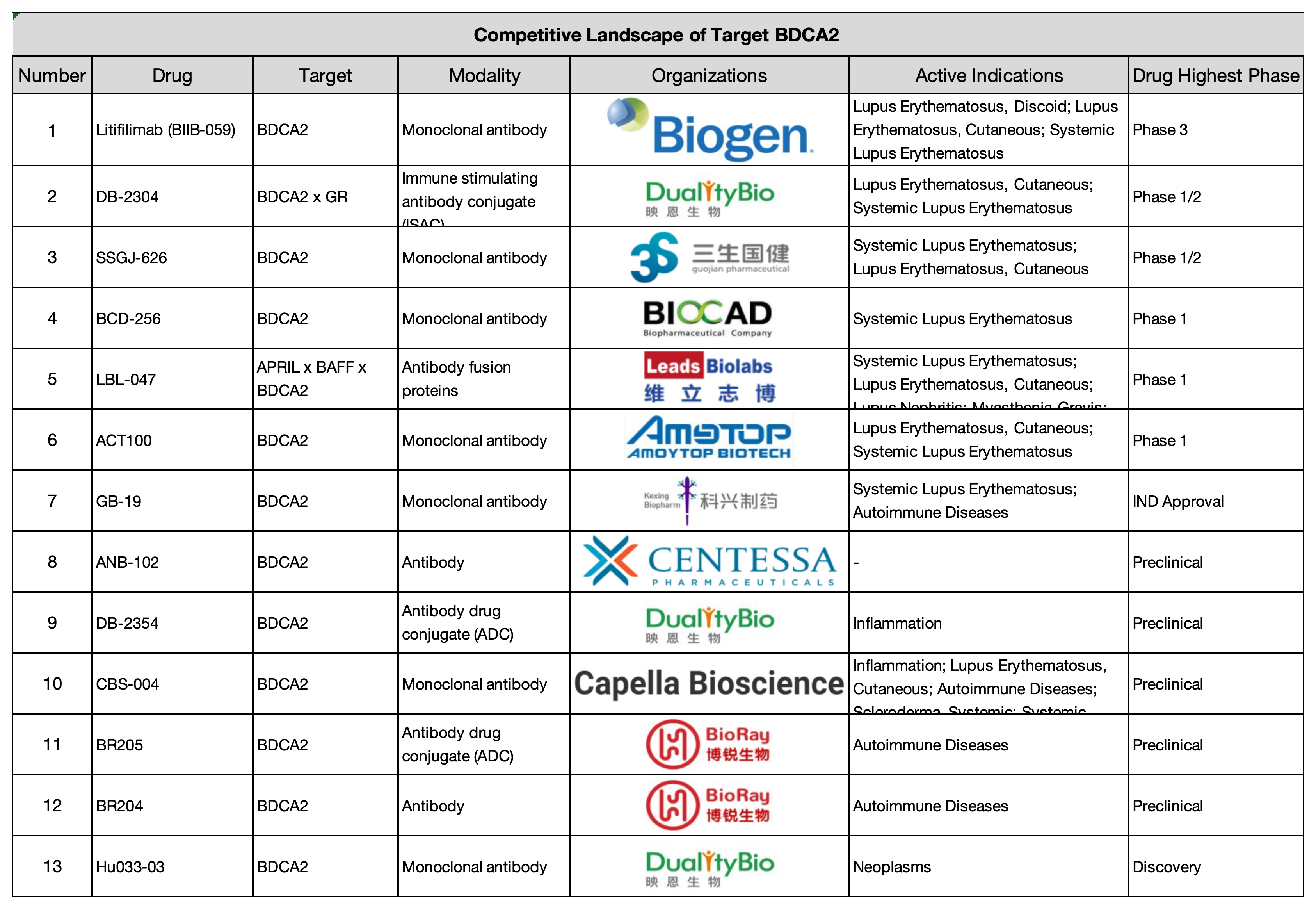
Global BDCA2 Research Pipeline