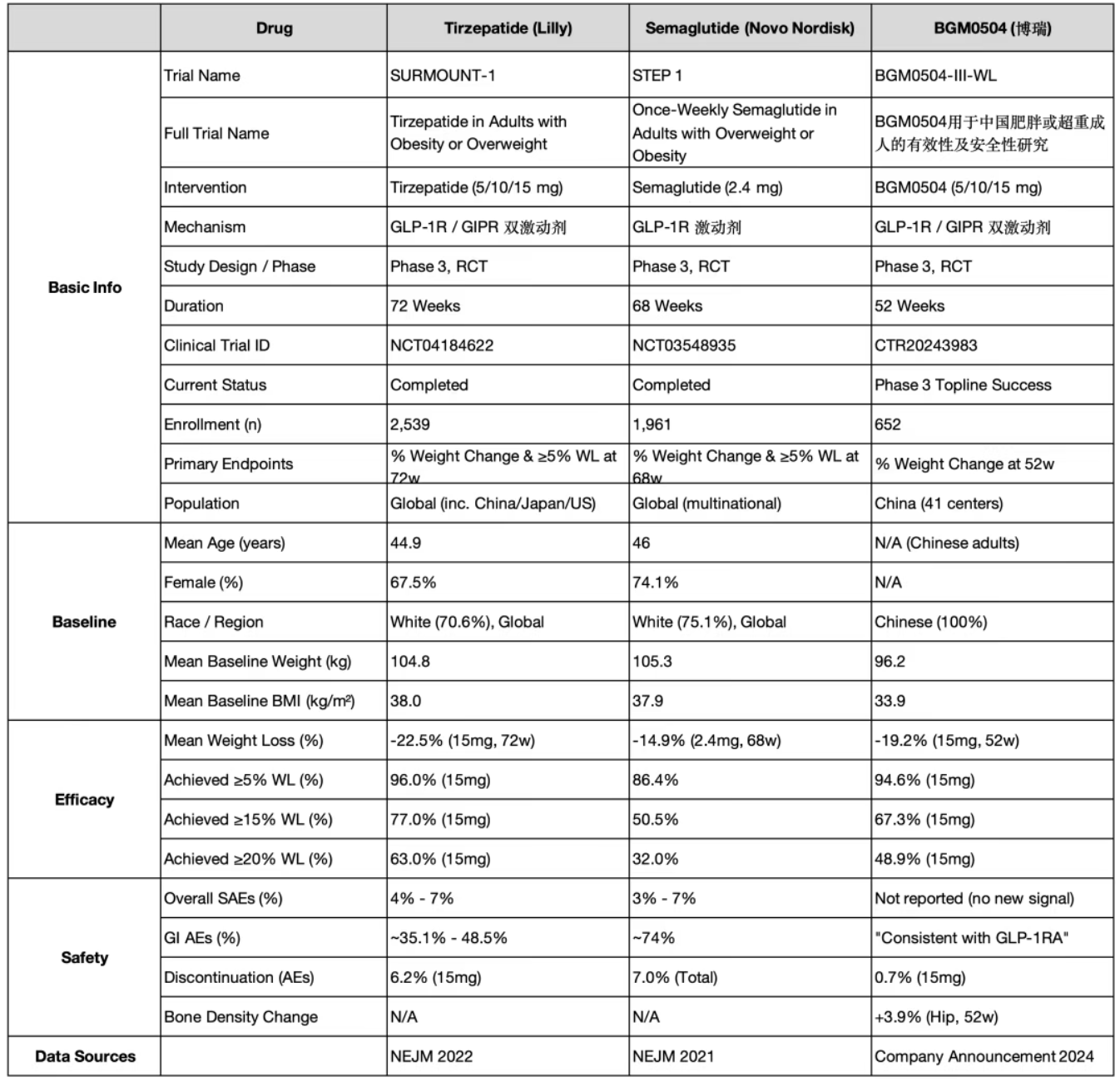
On May 5, BrightGene Bio-Medical (BrightGene) disclosed the Phase III clinical trial data for its self-developed GLP-1R/GIPR dual agonist, BGM0504 (Study: BGM0504-III-WL). The trial successfully met its primary and secondary endpoints.
To facilitate a benchmark comparison against Semaglutide and Tirzepatide, the author utilized the "Clinical Comparison Tool" by Yao Fanqie to perform a horizontal analysis of the classic clinical data for these three drugs.
Note: Since the China-specific clinical data for Tirzepatide has not yet been publicly released, the global multi-center data for Semaglutide and Tirzepatide were used for this comparison. Please consider potential geographic variations when reviewing.
Clinical Trials Referenced:
-
SURMOUNT-1 (NCT04184622): Evaluated the efficacy and safety of Tirzepatide (5/10/15 mg) in non-diabetic adults who are obese or overweight.
-
STEP 1 (NCT03548935): Evaluated the efficacy and safety of Semaglutide (2.4 mg) in non-diabetic adults who are obese or overweight.
-
BGM0504-III-WL (CTR20243983): Evaluated the efficacy and safety of BGM0504 (5/10/15 mg) in Chinese non-diabetic adults who are obese or overweight.
The comparison table is as follows: