
At the 2026 AACR Annual Meeting, D3 Bio unveiled the latest clinical trial data for its self-developed, next-generation KRAS G12C inhibitor, Elisrasib (D3S-001). Results demonstrate that Elisrasib possesses excellent anti-tumor activity and a favorable safety profile in patients with advanced non-small cell lung cancer (NSCLC), including both those who are KRAS-inhibitor naïve and those who have developed resistance to previous-generation drugs.
About Elisrasib
Developed independently by D3 Bio, Elisrasib operates on a principle similar to first-generation KRAS inhibitors: it selectively binds to the mutated KRAS G12C protein, "locking" it in an inactive state to cut off tumor growth signals. However, Elisrasib distinguishes itself through optimized molecular design, achieving faster and stronger target binding for more sustained and profound tumor suppression.
Clinical Data
The data presented at AACR stems from an ongoing Phase I/II clinical trial targeting KRAS G12C-mutant solid tumors. The study enrolled 165 patients with locally advanced or metastatic NSCLC who had previously received immunotherapy and/or chemotherapy (including a subset whose disease progressed after first-generation KRAS targeted therapy). Patients received Elisrasib orally once daily, with 600mg identified as the recommended therapeutic dose.
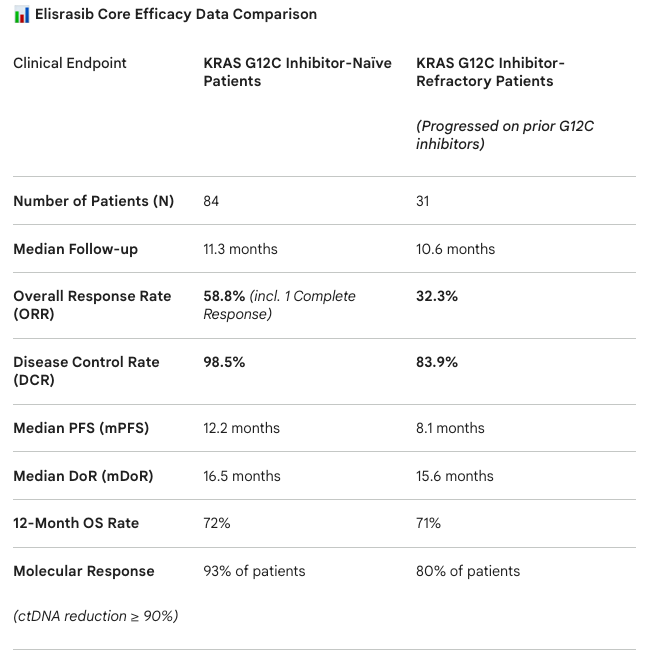
Key clinical outcomes include:
-
Clinical Benefit: Professor Cho noted that Elisrasib demonstrated significantly higher response rates and more durable tumor responses compared to first-generation inhibitors. Consequently, the FDA has granted Elisrasib both Fast Track and Breakthrough Therapy designations for the second-line treatment of KRAS G12C inhibitor-naïve patients.
-
Molecular Clearance: Analysis of circulating tumor DNA (ctDNA) revealed powerful molecular clearance. 93% of treatment-naïve patients and 80% of refractory patients saw a reduction in mutant ctDNA levels of over 90%.
-
Overcoming Resistance: Most encouragingly, the drug showed efficacy against KRAS gene amplification, a key mechanism of resistance to first-generation drugs. Among previously resistant patients, 5 cases of KRAS gene amplification were identified. Following Elisrasib treatment:
-
4 cases showed tumor shrinkage.
-
3 cases achieved clinical remission.
-
The Disease Control Rate (DCR) reached a perfect 100%.
-
-
4 cases showed tumor shrinkage.
-
3 cases achieved clinical remission.
-
The Disease Control Rate (DCR) reached a perfect 100%.
-
Safety Profile: The incidence of Grade 3 or higher treatment-related adverse events (TRAEs) was only 11.5%, demonstrating excellent safety and tolerability.
Conclusion
"Elisrasib has proven it can provide deeper and more lasting tumor responses, remaining effective even where first-generation KRAS G12C inhibitors have failed," summarized Professor Cho. "These findings suggest Elisrasib has the potential to significantly improve the treatment landscape for this patient population."
While these results from early-stage single-arm trials are promising, larger-scale randomized controlled trials will be necessary to further confirm long-term efficacy and safety.