On April 16, Eli Lilly and Company announced positive topline results from the Phase III ACHIEVE-4 trial of Foundayo (orforglipron), the first oral non-peptide GLP-1 receptor agonist. This marks the largest and longest study of Foundayo to date, enrolling more than 2,700 patients with type 2 diabetes (T2D) and elevated cardiovascular risk.
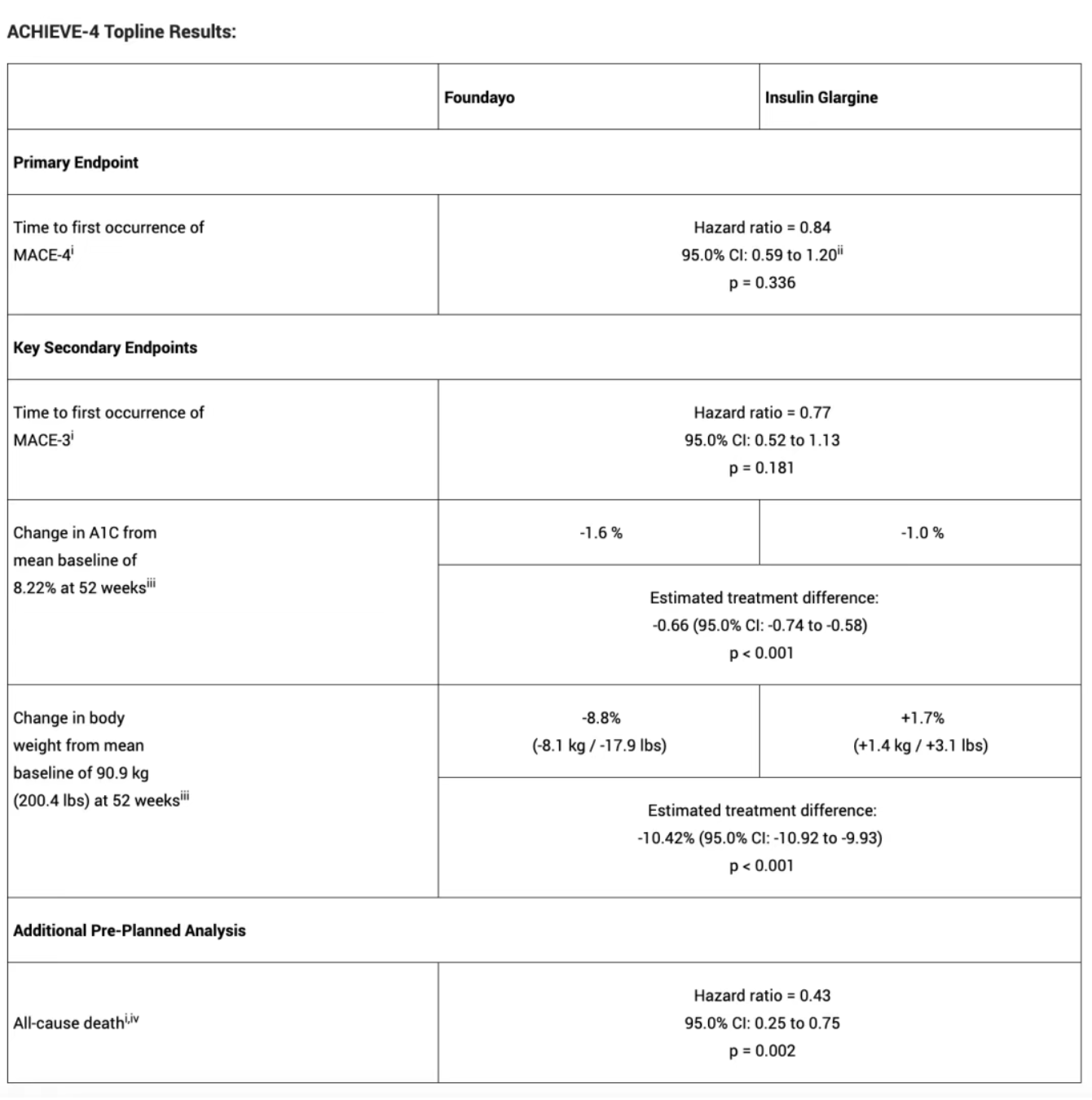
The study met its primary endpoint, demonstrating non-inferiority to insulin glargine in major adverse cardiovascular events (MACE-4; HR=0.84). A prespecified analysis showed a 57% reduction in all-cause mortality (HR=0.43). Foundayo also showed superior glycemic control (A1C difference −0.66%) and weight loss (−8.8% vs. +1.7% with insulin; difference −10.42%).
Eli Lilly plans to submit Foundayo for regulatory approval to the U.S. Food and Drug Administration for the treatment of T2D by the end of Q2, setting up direct competition with Novo Nordisk’s oral semaglutide (Wegovy).
ACHIEVE-4 Trial Overview
ACHIEVE-4 is a global, multicenter Phase III clinical trial conducted across 15 countries, enrolling over 2,700 patients with T2D who were overweight or obese and at high cardiovascular risk. The study aimed to evaluate the efficacy and safety of oral Foundayo compared with injectable insulin glargine.
Notably, the trial duration extended to approximately 104 weeks, making it the longest study in Foundayo’s clinical development program, designed to assess long-term cardiovascular and metabolic outcomes.
Key Results
1. 16% Reduction in Major Cardiovascular Events (MACE-4)
The trial successfully met its primary endpoint. Foundayo demonstrated non-inferiority to insulin glargine in reducing MACE-4 (cardiovascular death, myocardial infarction, stroke, or hospitalization for unstable angina). Patients treated with Foundayo showed a 16% lower risk of cardiovascular events (HR=0.84), meeting predefined non-inferiority criteria and confirming cardiovascular safety.
2. 57% Reduction in All-Cause Mortality
In a prespecified exploratory analysis, Foundayo reduced all-cause mortality by 57% compared with insulin glargine (HR=0.43). This substantial survival benefit in a high-risk population significantly strengthens the drug’s clinical value.
3. Superior Glycemic Control and Weight Loss
At Week 52, Foundayo demonstrated clear superiority over insulin glargine in both HbA1c reduction and weight loss. Patients on Foundayo achieved an average weight reduction of 8.8%, while those on insulin glargine experienced a 1.7% weight gain. This treatment difference was sustained through Week 104.
Additionally, Foundayo significantly improved multiple cardiovascular risk markers, including non-HDL cholesterol, systolic blood pressure, triglycerides, and high-sensitivity C-reactive protein (hsCRP), an important marker of inflammation.
Safety and Tolerability
The overall safety and tolerability profile of Foundayo was consistent with previous trials and aligned with known GLP-1 class effects. The most common adverse events were gastrointestinal, including nausea, vomiting, diarrhea, decreased appetite, and constipation.
Approximately 10.6% of patients discontinued treatment due to adverse events over at least 52 weeks, which falls within an acceptable range for this drug class.
Importantly, in response to prior regulatory concerns, ACHIEVE-4 included a dedicated analysis of drug-induced liver injury (DILI). The results showed no liver safety signals, consistent with findings from the ACHIEVE and ATTAIN programs, effectively addressing a key regulatory concern.
Summary and Outlook
On April 1, Foundayo received approval from the FDA for weight management, placing it firmly in the spotlight. The success of the ACHIEVE-4 trial further demonstrates its strong glycemic efficacy, long-term cardiovascular safety, and clean liver safety profile in T2D patients.
This positions Foundayo as a major contender in both diabetes and obesity markets. While semaglutide from Novo Nordisk has dominated due to its first-mover advantage, Foundayo’s once-daily oral formulation—without food or water restrictions—represents a significant breakthrough in convenience.
With its combined benefits in glucose control, weight loss, cardiovascular protection, and ease of use, Foundayo is poised to intensify competition with semaglutide, potentially reshaping the global metabolic disease treatment landscape.