Background

The treatment of low-grade upper tract urothelial cancer (LG UTUC) presents a significant challenge in balancing tumor eradication with kidney preservation. Padeliporfin Vascular Targeted Photodynamic Therapy (VTP) is an innovative drug-device combination. Patients receive an intravenous injection of the photosensitizer Padeliporfin, which is then activated locally by a near-infrared laser (753 nm) via endoscopy. This process selectively destroys tumor vasculature, leading to tumor necrosis and offering a potential organ-sparing treatment alternative.
Study Design
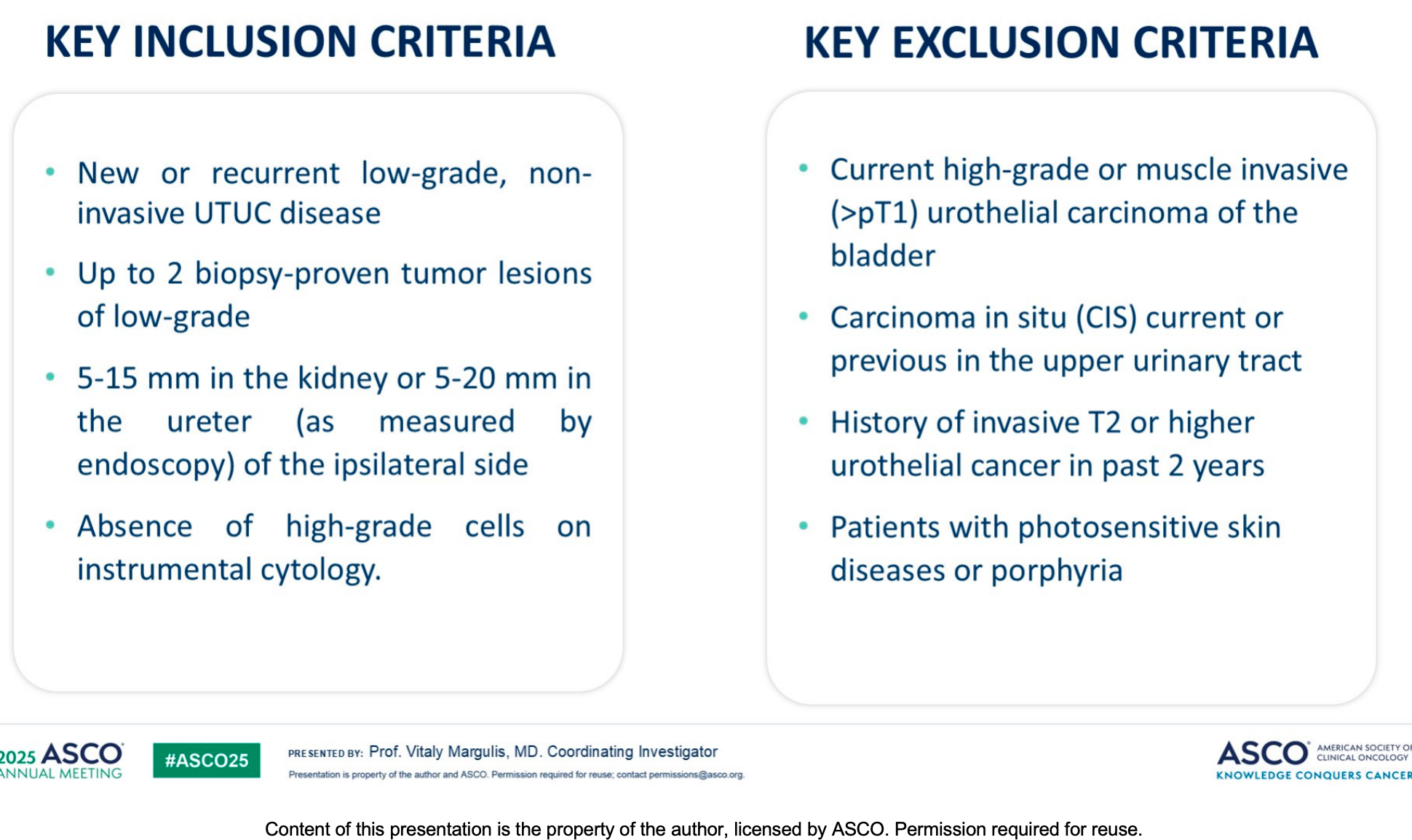
ENLIGHTED is a global, single-arm, non-randomized, open-label Phase 3 clinical trial evaluating the efficacy and safety of Padeliporfin VTP in treating LG UTUC. The study consists of an Induction Treatment Phase (ITP) and a Maintenance Treatment Phase (MTP). During the ITP, patients receive up to three VTP treatments at 4-week intervals until they achieve a Complete Response (CR) or experience treatment failure at the Primary Response Evaluation (PRE). Patients achieving CR enter the MTP for long-term endoscopic follow-up and repeat treatments if necessary.
Interim Efficacy Results
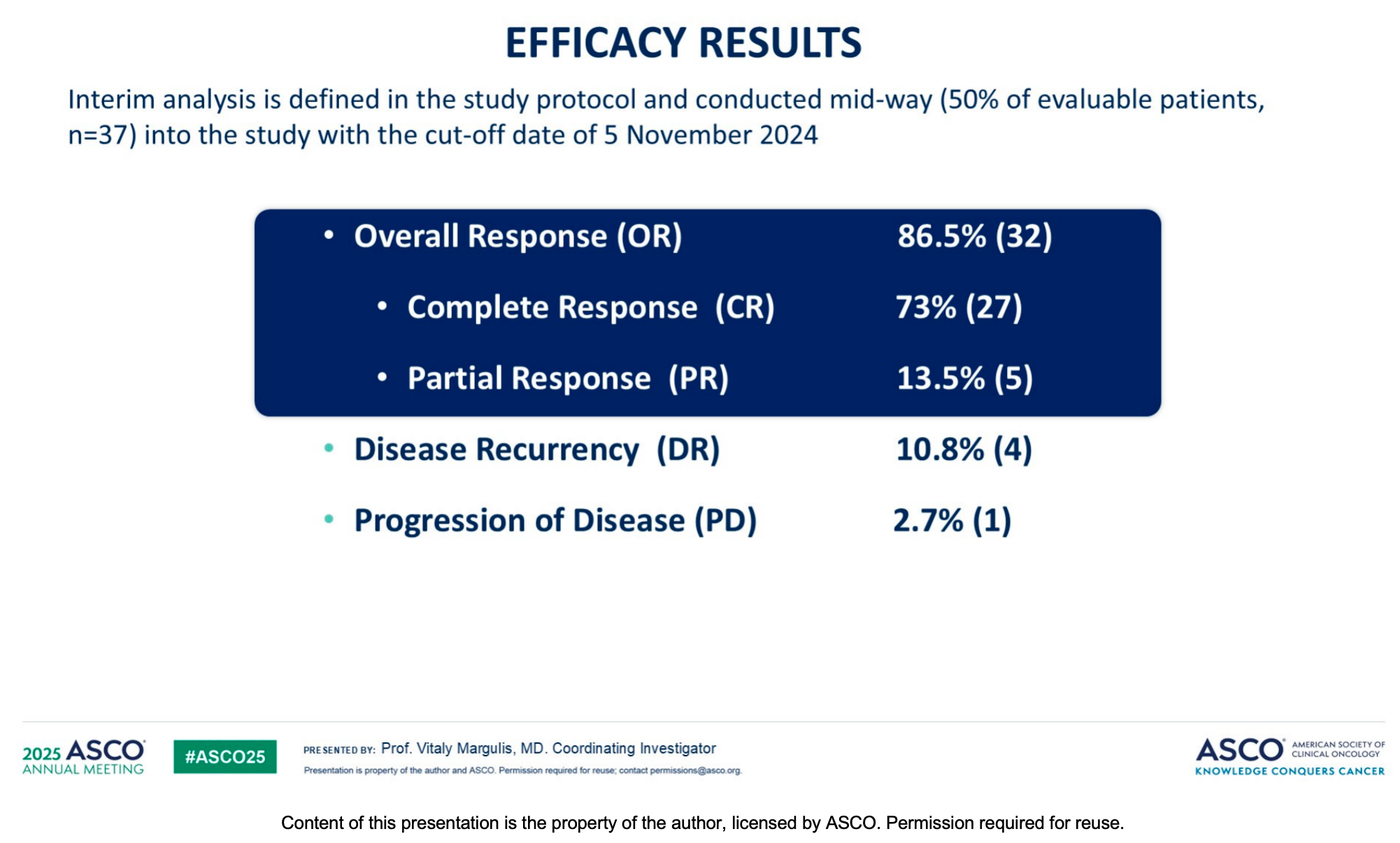
The interim data analysis demonstrates the outstanding clinical potential of Padeliporfin VTP. Among evaluable patients who completed the Induction Treatment Phase (ITP):
-
Complete Response (CR): Achieved in 70% to 73% of patients (based on the latest data cutoffs).
-
Partial Response (PR): Observed in approximately 13.5% to 18% of patients.
-
Overall Response Rate (ORR): Reached as high as 86.5% to 88%.
-
Recurrence and Progression: The disease recurrence rate was only 10.8%, and the progression rate was 2.7%.
Safety and Tolerability
Padeliporfin VTP demonstrated a favorable safety and tolerability profile, consistent with previous Phase 1 trial findings. The vast majority of treatment-emergent adverse events (TEAEs) were Grade 1-2 (mild to moderate), primarily including hematuria (11.6%-14%), flank pain (10%), procedural pain, dysuria, and nausea. These symptoms typically resolved within a few days. The incidence of Grade 3 serious adverse events (SAEs) was low (under 10%), mainly consisting of VTP-related renal colic and flank pain, which resolved rapidly within 1 to 2 days post-treatment. Notably, no irreversible complications such as significant ureteral strictures were observed.
Conclusion
The interim results from the ENLIGHTED Phase 3 study strongly support the potential of Padeliporfin VTP as a next-generation treatment for low-grade upper tract urothelial cancer (LG UTUC). The therapy not only demonstrated an exceptionally high complete response rate but also maintained an excellent safety profile. It holds great promise as a best-in-class treatment aimed at improving long-term patient outcomes and preserving kidney function.