On May 7th, U.S. biotech company Entrada Therapeutics announced positive top-line data from an early-stage clinical trial for ENTR-601-44, its investigational drug for the treatment of Duchenne Muscular Dystrophy (DMD).
Data from Entrada’s Phase 1/2 trial of ENTR-601-44 (an exon 44 skipping therapy) revealed that dystrophin levels increased by only 2.36%, far below the company’s original expectation of an increase exceeding 10%. Following the news, Entrada’s stock price plummeted 57% in pre-market trading that day.
About ENTR-601-44 and the EEV Platform
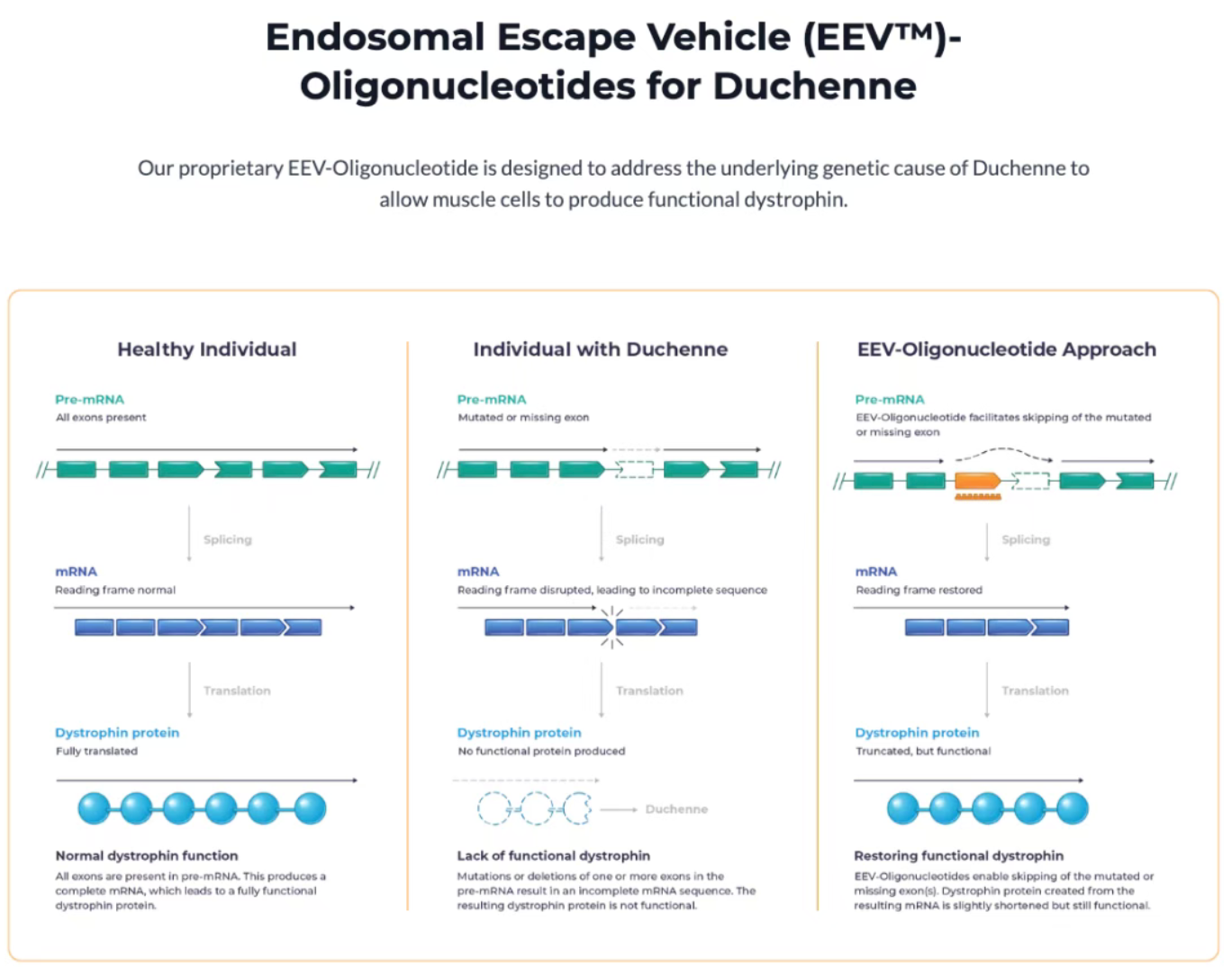
Duchenne Muscular Dystrophy (DMD) is a rare genetic muscle-wasting disease caused by mutations in the DMD gene. Due to a lack of dystrophin—the protein responsible for maintaining the structural integrity of muscle cells—patients experience progressive muscle weakness, often leading to premature death from heart failure or respiratory complications.
ENTR-601-44 is a proprietary Endosomal Escape Vehicle (EEV)-conjugated oligonucleotide drug developed by Entrada. It specifically targets DMD patients with mutations amenable to "Exon 44 skipping" (representing approximately 8% of the global DMD population). Its core mechanism involves helping the cellular splicing machinery "skip" defective exons, thereby producing a shorter but still partially functional dystrophin protein.
Clinical Data
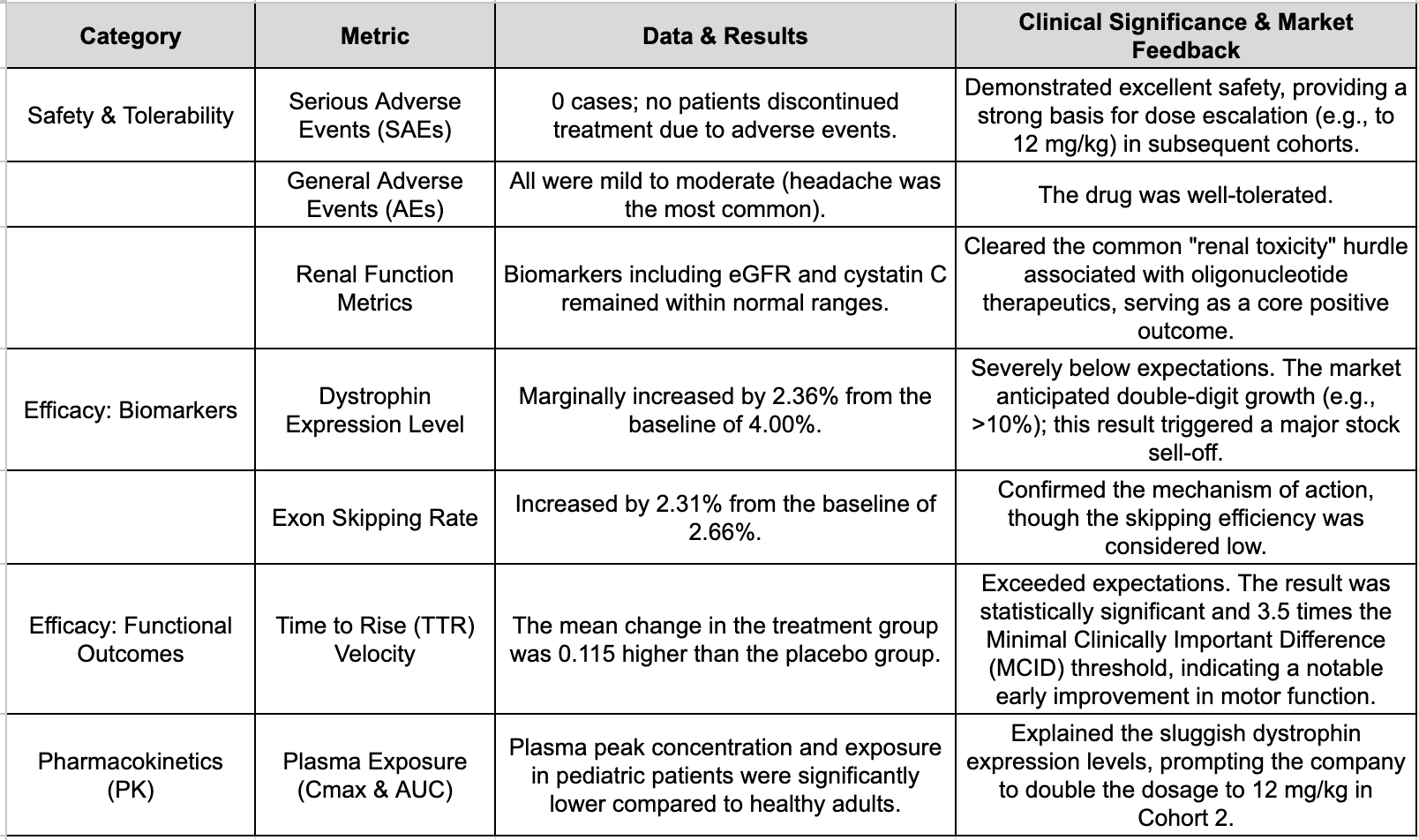
The data highlights from Cohort 1 of the ELEVATE-44-201 study (consisting of 8 patients aged 6–17 receiving a 6 mg/kg dose) focused on two main areas: safety and early improvements in motor function.
-
"Clean" Safety Profile: At the 6 mg/kg dose, there were no serious adverse events (SAEs), and no patients discontinued the trial. All treatment-emergent adverse events were mild to moderate (the most common being headaches). Crucially, renal function markers, including eGFR and Cystatin C, remained within normal ranges. Given that renal toxicity has historically been a significant hurdle for oligonucleotide drugs, this excellent safety and tolerability data is undoubtedly positive.
-
Surprising Early Functional Improvement: Despite being an early-stage trial with a short treatment duration, ENTR-601-44 demonstrated a statistically significant improvement in "Time to Rise (TTR) velocity," a key clinically validated metric. The average change in TTR velocity for the treatment group was 0.115 higher than the placebo group—a value 3.5 times the threshold for Minimal Clinically Important Difference (MCID). Company executives stated that this early functional improvement was both unexpected and highly clinically significant.
-
Dystrophin Expression Levels: According to the published data, treated patients showed a meager increase of only 2.36% in dystrophin levels (above a 4.00% baseline) in muscle tissue six weeks after administration. The exon skipping rate also increased by only 2.31% over a 2.66% baseline. These figures fell significantly short of market and company expectations. Analysts at investment bank William Blair noted that Wall Street’s baseline expectation for this metric was an increase of over 10%, while Entrada management had previously guided toward "double-digit increases."
The contrast with competitors intensified the blow. Analysts pointed out that del-zota, an oligonucleotide therapy from competitor Avidity Biosciences, showed an approximately 25% increase in dystrophin after one year of treatment in previous readouts (though no rigorous head-to-head comparison exists). In a competitive "Best-in-Class" landscape, a single-digit increase of 2.36% was clearly insufficient for demanding investors.
Reasons Behind the Data Discrepancy: PK Differences Between Children and Adults
Addressing the lower-than-expected protein expression, Entrada offered a scientific explanation: significant differences in pharmacokinetics (PK) between pediatric patients and healthy adults.
Data showed that the peak plasma concentration ($C_{max}$) and the area under the plasma concentration-time curve ($AUC$) for pediatric patients in Cohort 1 were much lower than those observed in previous healthy adult volunteers. Entrada CEO Dipal Doshi admitted to being surprised by the discrepancy but noted that the phenomenon aligns closely with recent data the company obtained from juvenile non-human primates (NHPs). In short, insufficient plasma exposure led to sluggish protein expression.
Outlook
Despite the stock market "Waterloo," Entrada’s clinical progress has not been halted. The silver lining is that the excellent safety profile shown in Cohort 1 paves the way for higher dosing. The study has already initiated Cohort 2, which doubles the dose to 12 mg/kg. The company's latest PK models predict that at 12 mg/kg, the plasma $AUC$ will increase significantly, thereby driving a substantial rise in dystrophin expression levels.