Background and Study Design

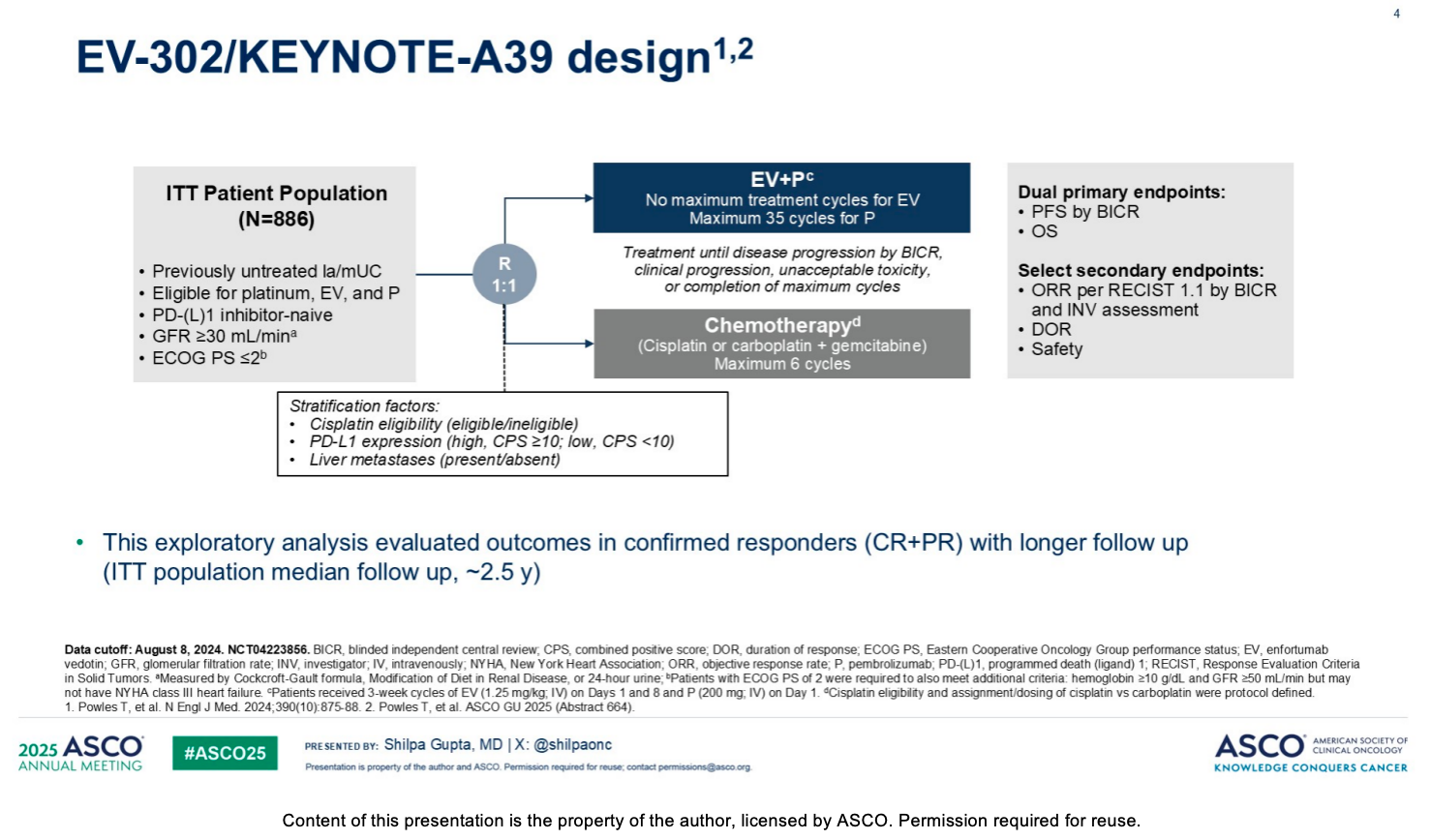
In the global randomized phase 3 EV-302 study targeting patients with previously untreated locally advanced or metastatic urothelial carcinoma (la/mUC), enfortumab vedotin (EV) plus pembrolizumab (P) has been established as the standard of care for first-line treatment. Previous data showed that in a broad patient population, EV+P demonstrated superior efficacy compared with platinum-based chemotherapy, with a median progression-free survival (PFS) of 12.5 months (vs. 6.3 months in the chemotherapy arm) and a median overall survival (OS) of 33.8 months (vs. 15.9 months in the chemotherapy arm). This updated exploratory analysis focuses on responders who achieved a confirmed response (i.e., patients who achieved a complete response [CR] or partial response [PR]), with a median follow-up of approximately 2.5 years (29.1 months). The study utilized a 1:1 randomization to evaluate EV+P (no maximum treatment cycles for EV, maximum of 35 cycles for P) compared to a maximum of 6 cycles of chemotherapy.
Core Efficacy Data and Response Rates
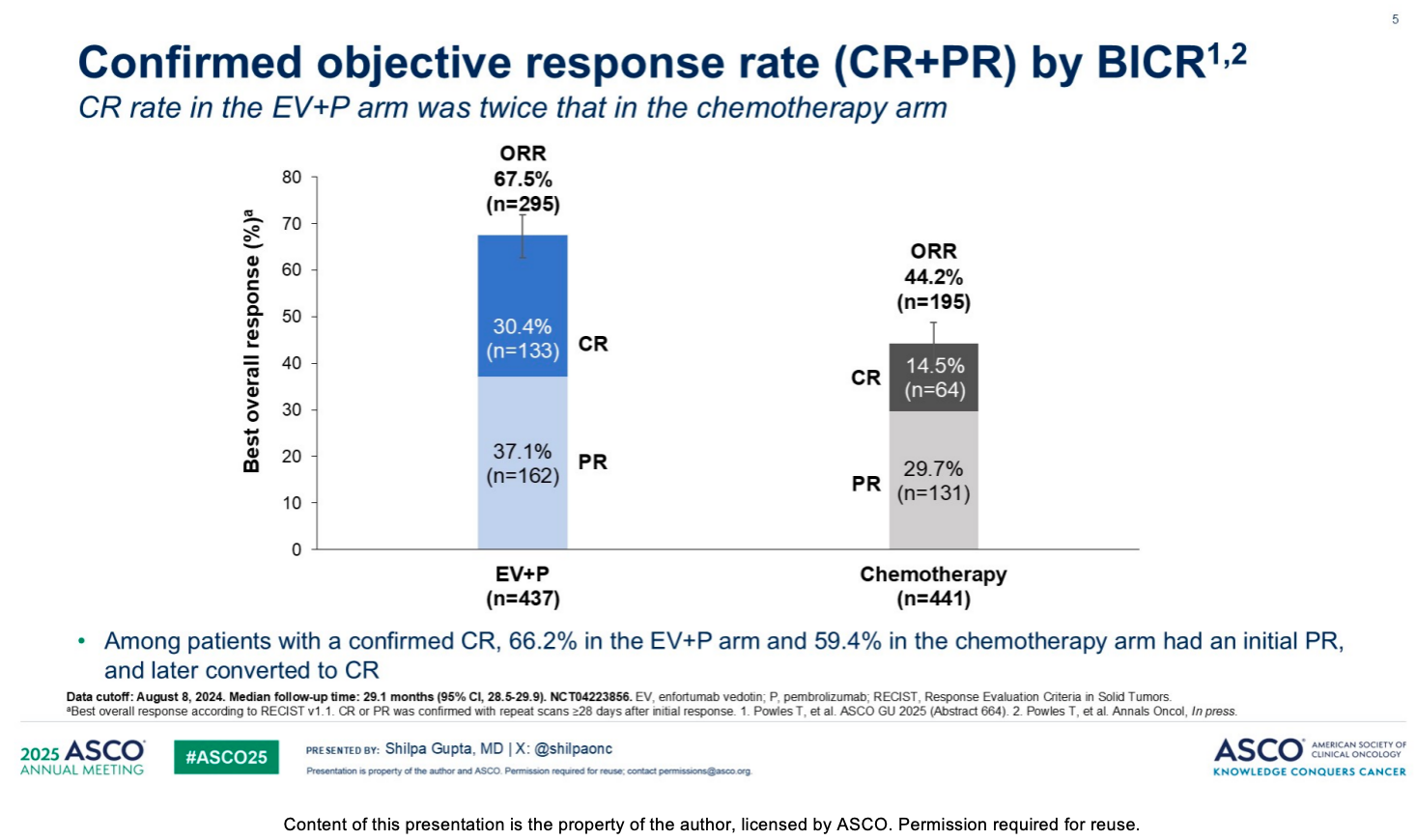
Among patients who achieved a response, the EV+P regimen demonstrated a doubled capacity for deep response compared to traditional chemotherapy. The objective response rate (ORR) in the EV+P arm reached 67.5% (n=295), whereas the ORR in the chemotherapy arm was 44.2% (n=195). Notably, the complete response (CR) rate in the EV+P arm was 30.4%, which is more than twice that of the chemotherapy arm (14.5%). Among patients with a confirmed complete response (CR), 66.2% in the EV+P arm (and 59.4% in the chemotherapy arm) had an initial partial response (PR) that later converted to a CR. There were more responders in the EV+P arm than in the chemotherapy arm across all baseline characteristic subgroups, including age (<65 or ≥65 years), ECOG performance status, primary tumor location, metastatic category (such as visceral metastases or lymph node-only disease), and cisplatin eligibility status.
Duration of Response (DOR)
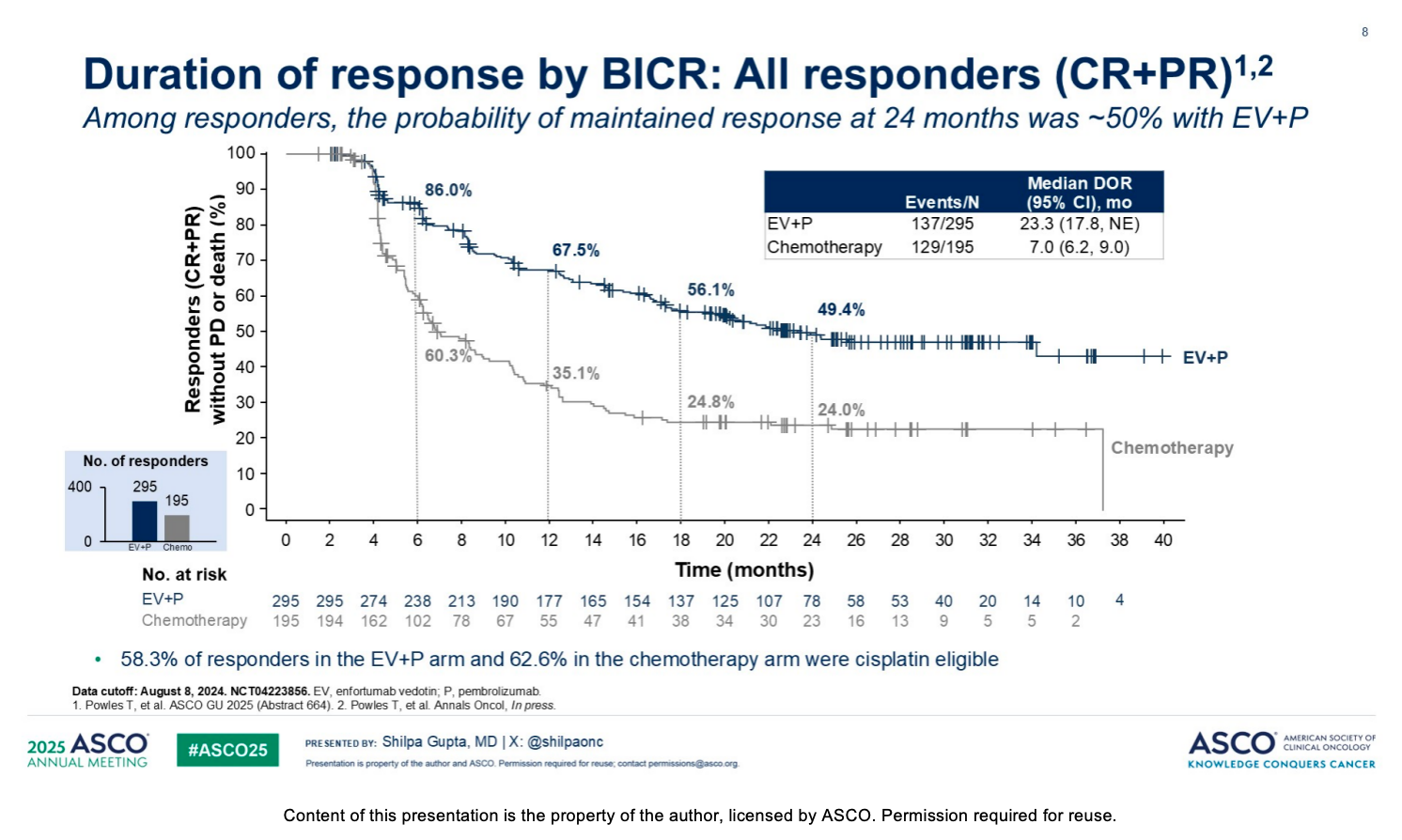
The EV+P regimen not only improved response rates but also significantly prolonged the duration of disease control for responders. Assessment by blinded independent central review (BICR) showed that the median duration of response (DOR) for responders in the EV+P arm reached 23.3 months, compared to only 7.0 months in the chemotherapy arm. At 24 months, the probability of maintaining a response among responders in the EV+P arm was approximately 50% (specifically 49.4%). Furthermore, responses to the EV+P regimen were highly durable for patients who achieved a complete response (CR), with a 74% probability of maintained CR at 24 months.
Safety and Dose Modifications
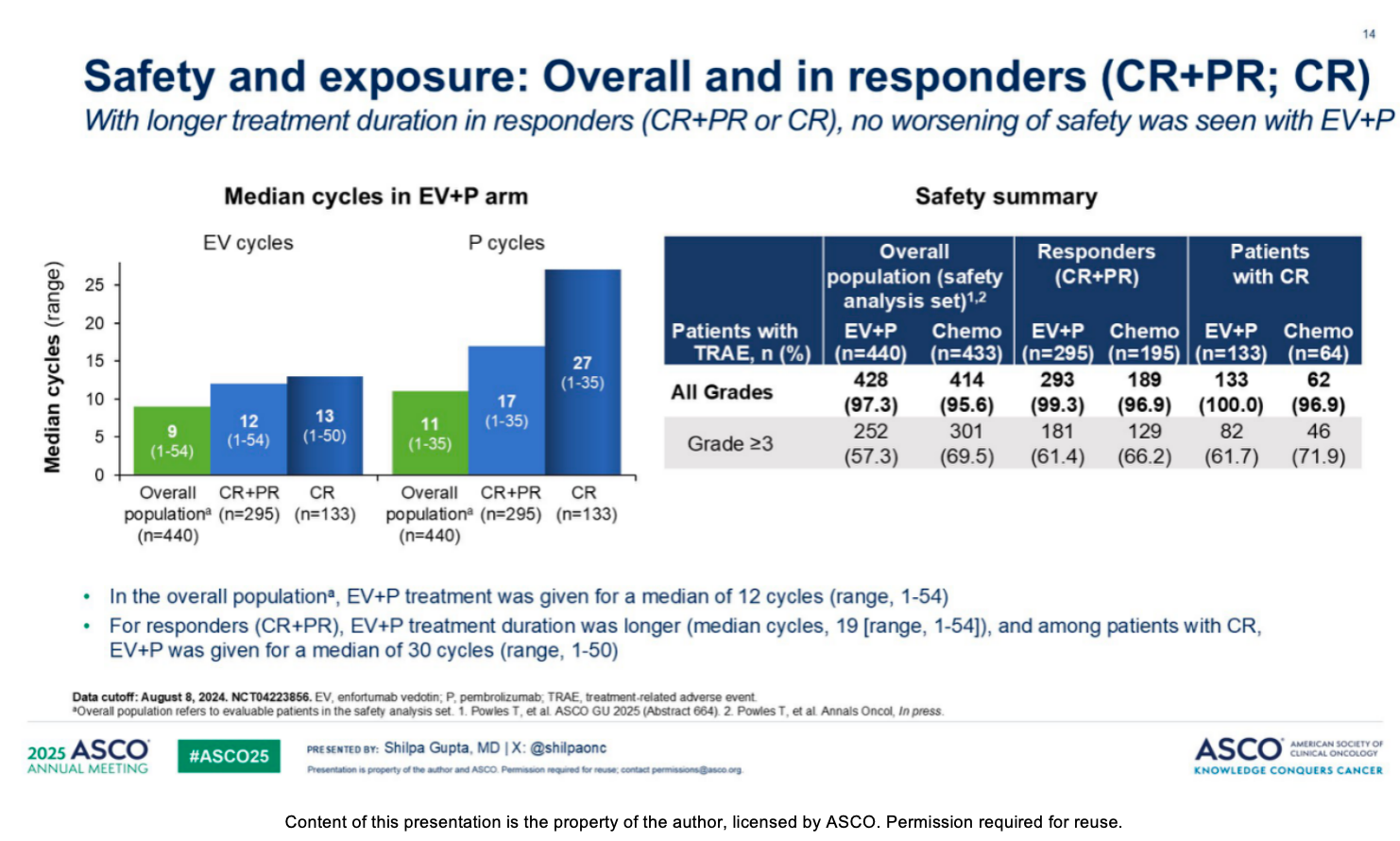
Although responders received treatment for a longer duration, the overall safety profile of EV+P remained manageable and was consistent with the overall population. In the EV+P arm, the median EV treatment duration for responders was 9.7 months (compared to 7.1 months for the overall population), and the median pembrolizumab treatment duration was 13.2 months (compared to 8.5 months for the overall population). The incidence and severity of adverse events of special interest for both EV and pembrolizumab (such as peripheral sensory neuropathy, rash, severe skin reactions, and hyperglycemia) among responders were generally consistent with the overall population. Due to the longer treatment duration, dose modifications resulting from treatment-related adverse events (TRAEs) were more common among responders. Among them, 69.2% of responders experienced a dose interruption (compared to 59.8% in the overall population), and 53.9% experienced an EV dose reduction (compared to 43.0% in the overall population). Appropriate dose modifications and interruptions allowed responders to continue treatment, maintaining a safe tolerability profile despite receiving more cycles of therapy.
Conclusion

This exploratory analysis demonstrates that EV+P improved survival compared to chemotherapy in patients who achieved a CR or PR. The EV+P regimen not only achieved a complete response rate twice that of chemotherapy, but the responses were also durable. Collectively, these data reinforce EV+P as the standard of care for the first-line treatment of la/mUC, irrespective of baseline characteristics, including cisplatin eligibility