Introduction
For patients with unresectable or metastatic urothelial carcinoma (mUC), the medical community has been actively exploring more effective and durable first-line treatment options. At the 2025 Annual Meeting of the American Society of Clinical Oncology (ASCO), investigators presented the highly anticipated final results of the CheckMate 901 trial (NCT03036098).
This report specifically focuses on patients who are ineligible for cisplatin-based chemotherapy, evaluating the clinical performance of a dual immunotherapy regimen—nivolumab (Opdivo, “O”) plus ipilimumab (Yervoy, “Y”)—compared with the standard gemcitabine plus carboplatin (Gem-Carbo) chemotherapy.
Study Design and Baseline Characteristics
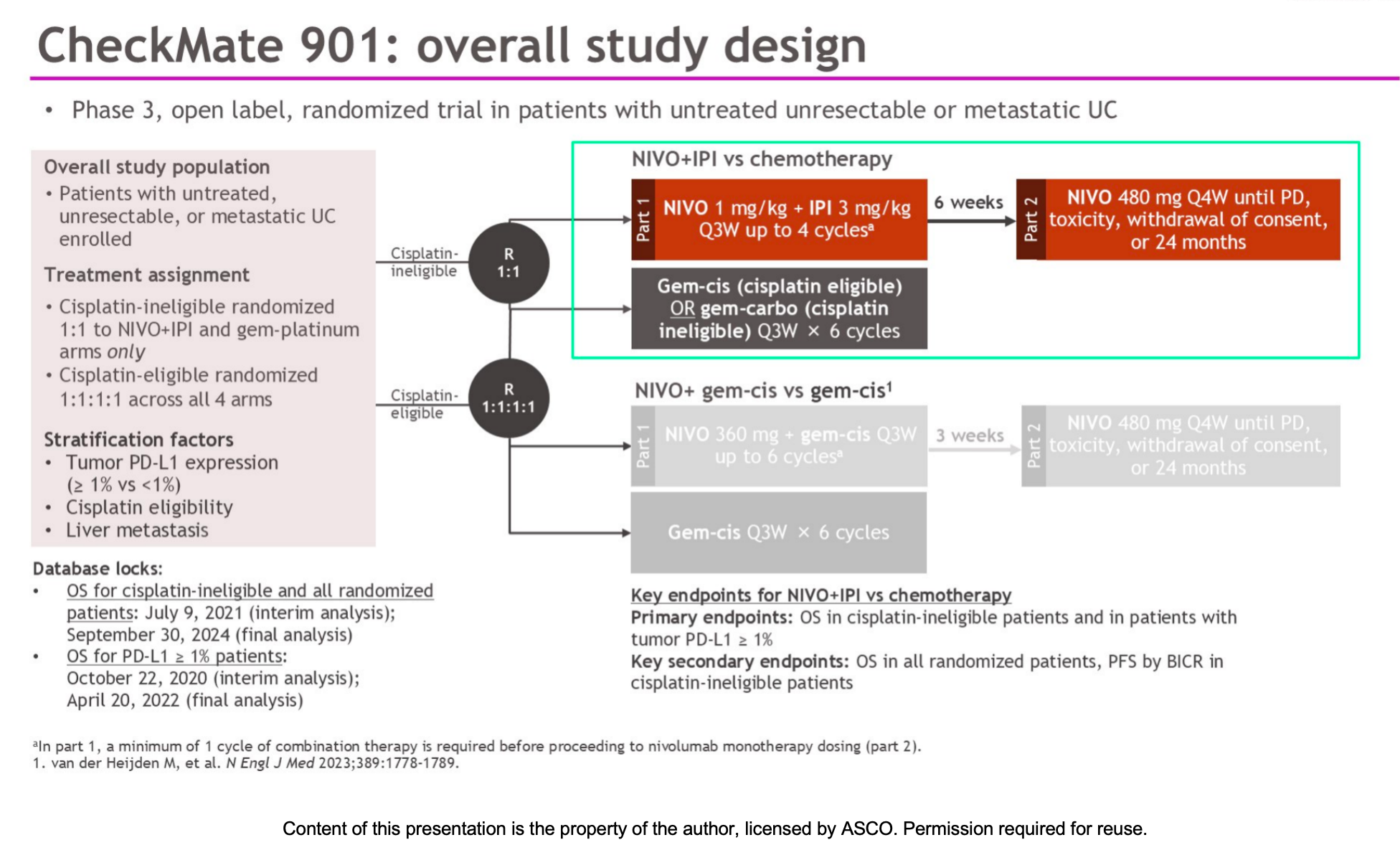
CheckMate 901 is a global, open-label, phase 3 randomized clinical trial. In this specific cohort, a total of 445 previously untreated patients with metastatic or unresectable urothelial carcinoma who were ineligible for cisplatin (glomerular filtration rate between 30 and <60 mL/min) were enrolled.
Patients were randomized in a 1:1 ratio into two groups:
- Immunotherapy Combination Group (n=221):
Nivolumab (1 mg/kg) plus ipilimumab (3 mg/kg) administered every 3 weeks for up to 4 cycles, followed by nivolumab monotherapy maintenance until disease progression or unacceptable toxicity (maximum of 2 years). - Chemotherapy Group (n=224):
Standard gemcitabine plus carboplatin chemotherapy.
The primary endpoint was overall survival (OS). Secondary and exploratory endpoints included progression-free survival (PFS), objective response rate (ORR), duration of response (DOR), and safety.
Key Efficacy Results: No Statistical Significance, but Meaningful Long-Term Benefit
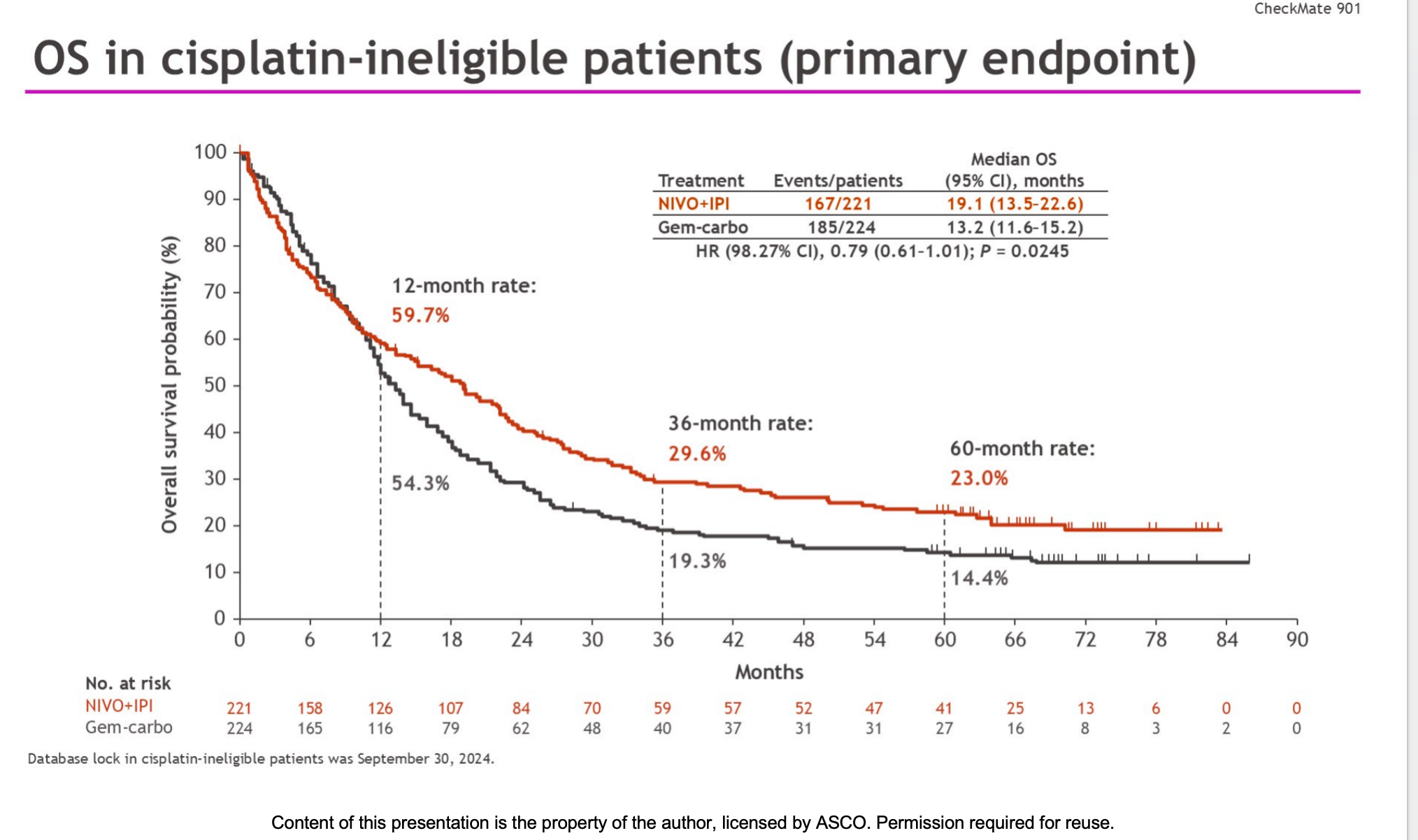
After a median follow-up of 58.3 months, the results revealed distinct characteristics between the two treatment approaches:
- Overall Survival (OS):
Median OS was 19.1 months in the immunotherapy group versus 13.2 months in the chemotherapy group (HR = 0.79 [98.27% CI, 0.61–1.01]; P = 0.0245).
Unfortunately, this improvement did not meet the pre-specified threshold for statistical significance. - Objective Response and Duration of Response (DOR):
ORR was similar between the two groups (35.3% vs 38.8%). However, immunotherapy demonstrated a substantial advantage in durability. The median DOR in the immunotherapy group reached 25.0 months—more than three times longer than that in the chemotherapy group (7.4 months). - Long-Term Survival (Landmark OS):
At 36 months, the OS rate was 29.6% in the immunotherapy group, compared with 19.3% in the chemotherapy group.
Safety and Tolerability
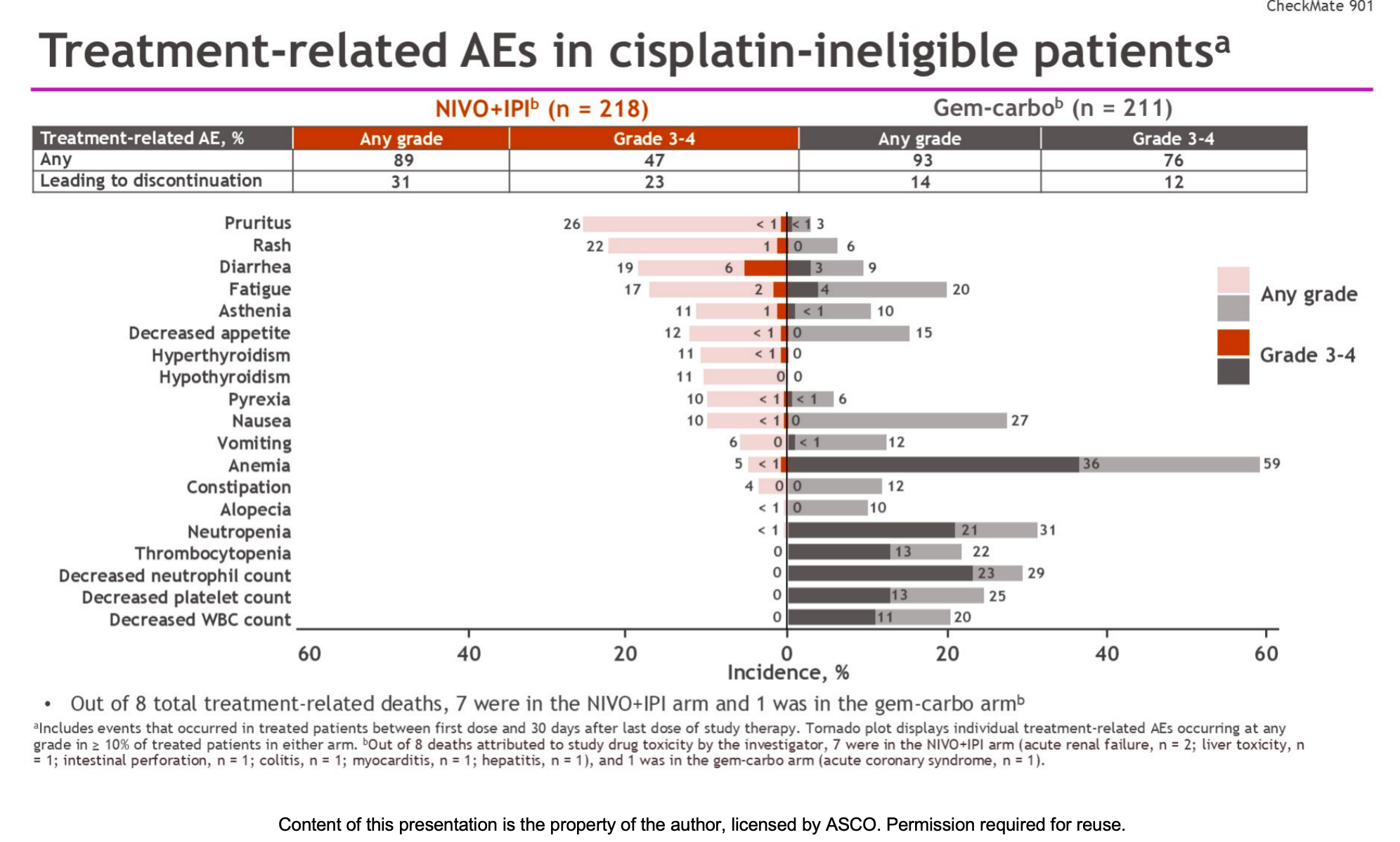
No new safety signals were identified.
- The incidence of treatment-related adverse events (TRAEs) of any grade was 89.0% in the immunotherapy group, with 47.2% being grade 3–4 events.
- In the chemotherapy group, TRAEs of any grade occurred in 92.9% of patients, with a significantly higher rate of grade 3–4 events (76.3%), primarily driven by hematologic toxicity.
- However, treatment discontinuation due to adverse events was higher in the immunotherapy group (31.2% vs 14.2%), mainly due to immune-related adverse events.
Conclusion and Clinical Implications
In the first-line treatment of cisplatin-ineligible advanced urothelial carcinoma, nivolumab plus ipilimumab did not reach statistical significance for overall survival. Nevertheless, it delivered clinically meaningful benefits, particularly in terms of durable responses (median DOR of 25 months) and improved long-term survival rates.
These findings suggest that, for a subset of patients, this limited-duration, chemotherapy-free dual immunotherapy regimen offers substantial antitumor activity.
Future research should focus on identifying predictive biomarkers to better select patients who are most likely to achieve long-term survival benefits from this dual immunotherapy approach.