On May 11, clinical-stage biotechnology company Fractyl Health announced that Dutch regulators had authorized the initiation of a Phase 1/2 first-in-human clinical trial for its lead pipeline candidate, RJVA-001. The therapy has become the world’s first gene therapy for type 2 diabetes (T2D) to enter clinical development. By delivering the GLP-1 gene directly to the pancreas, RJVA-001 aims to provide a “one-time, long-term remission” solution for metabolic diseases. The company expects to dose the first patient in the second half of 2026 while simultaneously advancing the platform into obesity treatment.
Mechanism of Action and Clinical Trial Design
1. Innovative MOA: Rejuva® Smart GLP-1™ Platform and Localized Delivery Technology
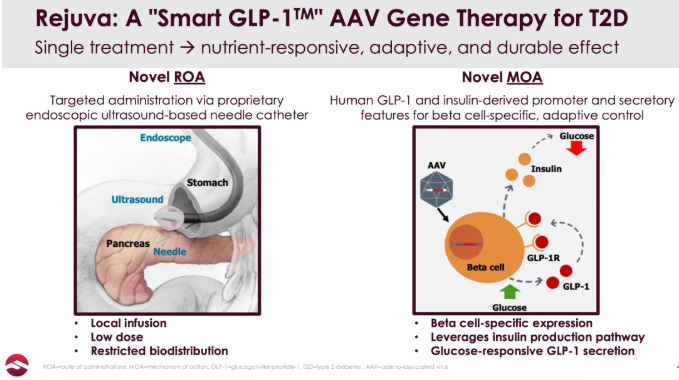
For drug development experts, one of the greatest technical challenges in applying AAV gene therapy to widespread chronic metabolic diseases is balancing tissue specificity with systemic safety.
RJVA-001 is built on Fractyl’s proprietary Rejuva® platform. Instead of conventional intravenous infusion, the therapy uses endoscopic ultrasound-guided (EUS) delivery to administer the treatment directly into the pancreas. This localized administration approach significantly reduces systemic exposure of AAV vectors to off-target organs such as the liver, thereby improving safety.
Mechanistically, RJVA-001 specifically targets pancreatic beta cells. Through vector-mediated gene delivery, pancreatic islet cells gain the ability to synthesize and secrete GLP-1 autonomously. More importantly, its promoter has been specially engineered to trigger physiological, pulsatile GLP-1 release after meals (“Smart GLP-1”), mimicking the body’s natural hormonal regulation pathways. This differs fundamentally from current GLP-1 receptor agonists (GLP-1RAs), which rely on exogenous systemic administration and often maintain sustained supraphysiological GLP-1 levels.

2. Phase 1/2 First-in-Human (FIH) Trial Design
- Trial Type: Open-label, multicenter, single ascending dose (SAD) study.
- Target Population: Adults aged 35–70 years with BMI 27–40 kg/m² and inadequately controlled T2D (HbA1c 7.0%–10.0%) despite treatment with GLP-1RAs and up to three non-insulin oral antidiabetic drugs.
- Dose and Cohorts: Three escalating dose cohorts (3 patients each), followed by an expansion cohort of up to 20 patients.
- Clinical Endpoints: Patients will undergo 12 months of primary follow-up to evaluate safety, tolerability, glycemic control, immune response, and pancreatic beta-cell function, followed by up to 5 years of long-term monitoring to assess durability of the gene therapy.
Milestone Significance
1. Opening the Era of Gene Therapy for Chronic Metabolic Diseases
Historically, gene therapy has primarily focused on rare diseases, monogenic disorders such as spinal muscular atrophy (SMA) and hemophilia, as well as oncology. RJVA-001, as the first clinical-stage AAV gene therapy for T2D, marks the expansion of gene delivery technologies into highly prevalent and complex chronic metabolic diseases affecting hundreds of millions of patients worldwide.
If successful, this approach could transform metabolic disease treatment from lifelong symptom management into a one-time, organ-level disease-modifying intervention, potentially overcoming long-standing patient adherence challenges.
2. Establishing Fractyl Health’s “Dual Clinical-Stage Platform” Position
The approval is also strategically important for Fractyl Health itself. The company currently operates two core technology platforms:
- Revita® — a duodenal mucosal resurfacing platform currently in pivotal clinical development (REMAIN-1) for weight maintenance after GLP-1 discontinuation.
- Rejuva® — a pancreatic gene therapy platform that officially entered clinical-stage development with RJVA-001.
This milestone significantly broadens Fractyl’s pipeline portfolio and may strengthen its valuation and partnership potential with multinational pharmaceutical companies.
Outlook
Following approval of the Dutch Clinical Trial Application (CTA), Fractyl Health has outlined an ambitious development roadmap:
- Clinical Progress and Data Readouts: The company plans to administer the first RJVA-001 dose in the second half of 2026, subject to site activation timelines, with preliminary clinical data expected later that year.
- Global Multicenter Expansion: Fractyl has also submitted a clinical trial application in Australia and expects regulatory feedback in Q3 2026 to accelerate Asia-Pacific and global clinical expansion.
- Next-Generation Pipeline Development: Beyond RJVA-001 for T2D, the Rejuva platform is advancing RJVA-002, a preclinical dual GIP/GLP-1 agonist AAV gene therapy targeting the rapidly growing obesity market.