In the treatment landscape of advanced urothelial carcinoma (UC), first-line (1L) maintenance strategies remain a central focus for the industry. In newly released research data, the interim analysis of the JAVELIN Bladder Medley phase 2 trial provides robust evidence-based support for the cutting-edge strategy of combining immunotherapy with an antibody-drug conjugate (ADC). This study rigorously evaluated the efficacy and safety of avelumab plus sacituzumab govitecan (SG) versus avelumab monotherapy as a 1L maintenance treatment for patients with advanced UC.
Below is a structured synthesis and in-depth analysis of the core findings from this clinical trial.
I. Executive Summary
Significant PFS Benefit: In patients with advanced UC who did not progress after 1L platinum-based chemotherapy (PBC), maintenance treatment with avelumab + SG significantly improved progression-free survival (PFS) compared to avelumab monotherapy, achieving a stratified hazard ratio (HR) of 0.49 [95% CI, 0.31-0.76].
Manageable Safety Profile: Although adverse events with the combination therapy increased, they were consistent with the known safety profiles of each individual drug, with no new safety signals identified.
Strategic Outlook: Combining avelumab with anti-Trop-2 ADCs represents a highly promising strategy to improve patient outcomes in advanced UC.
II. Background & Rationale
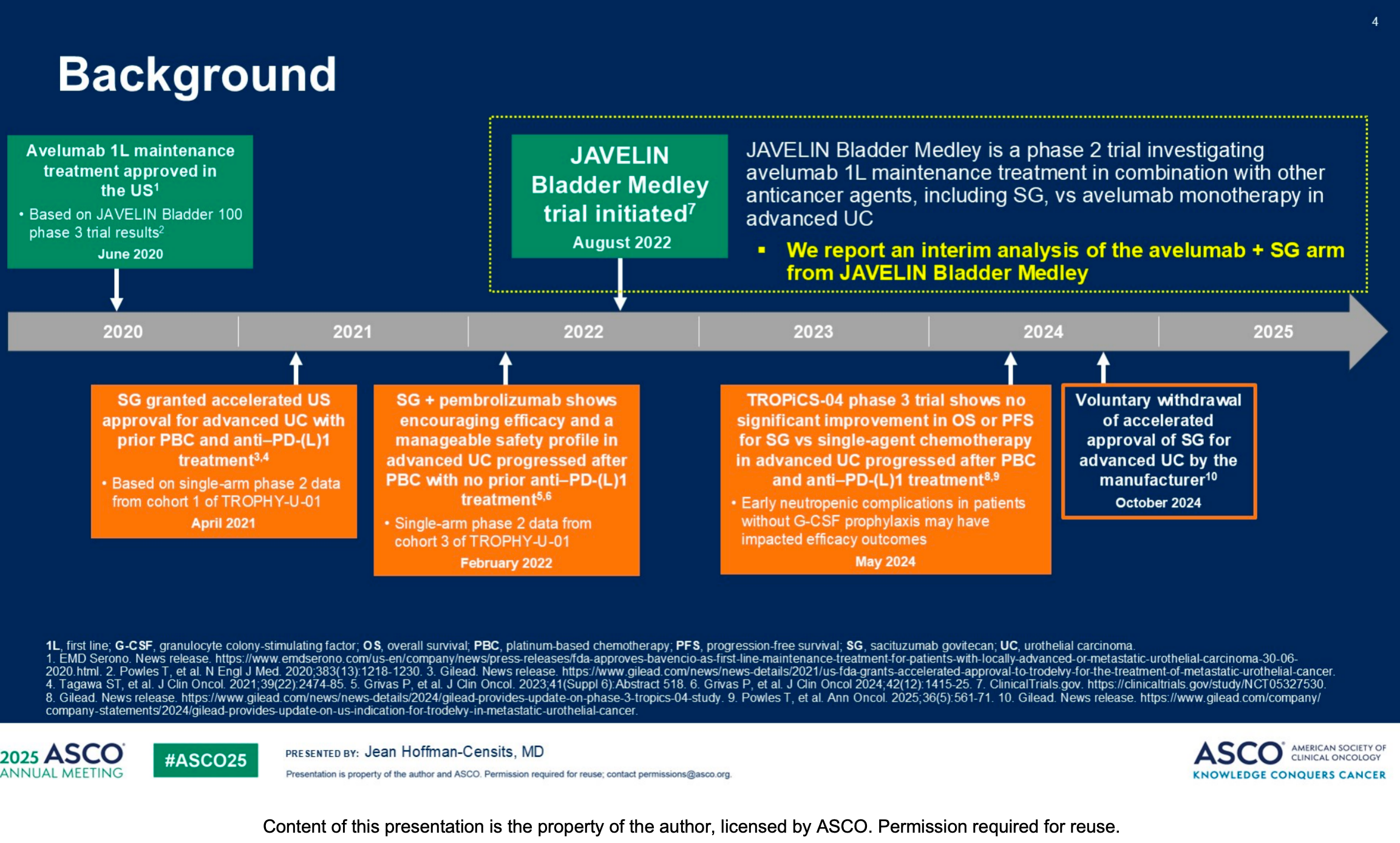
Avelumab 1L maintenance is currently the recommended standard of care for patients with advanced UC who have not progressed following 1L PBC. This clinical foundation was established by the landmark JAVELIN Bladder 100 phase 3 trial.
However, to further break through the survival benefit bottlenecks of existing therapies, the industry has begun exploring combinations of immune checkpoint inhibitors with drugs utilizing different mechanisms. Sacituzumab govitecan (SG), a Trop-2-targeted ADC, has thus entered the researchers' field of vision.
Against this backdrop, the JAVELIN Bladder Medley trial was initiated to investigate avelumab 1L maintenance treatment in combination with other anticancer agents, including SG, in advanced UC. This disclosure focuses on the interim analysis of the avelumab + SG arm.
III. Innovative Trial Design & Methodology

JAVELIN Bladder Medley is an international, randomized, open-label phase 2 trial.
Eligibility Criteria: Patients with unresectable locally advanced or metastatic UC who achieved no progressive disease (PD) after 4-6 cycles of 1L PBC (cisplatin and/or carboplatin + gemcitabine), with an ECOG performance status of 0-1.
Trial Endpoints: This interim analysis was triggered by PFS events. Primary endpoints are PFS and safety; secondary endpoints encompass overall survival (OS), objective response, and duration of response.
Innovative Statistical Design: To enhance statistical power, the study incorporated external data into the control arm design. Per protocol, PFS and OS data in the control arm were extended using external data from the JAVELIN Bladder 100 phase 3 trial. To account for population differences, external patients were propensity-score weighted using predefined prognostic factors, and the sum of external patients was downweighted to 37 to be equal to the number of randomized patients.
IV. Core Efficacy & Safety Data
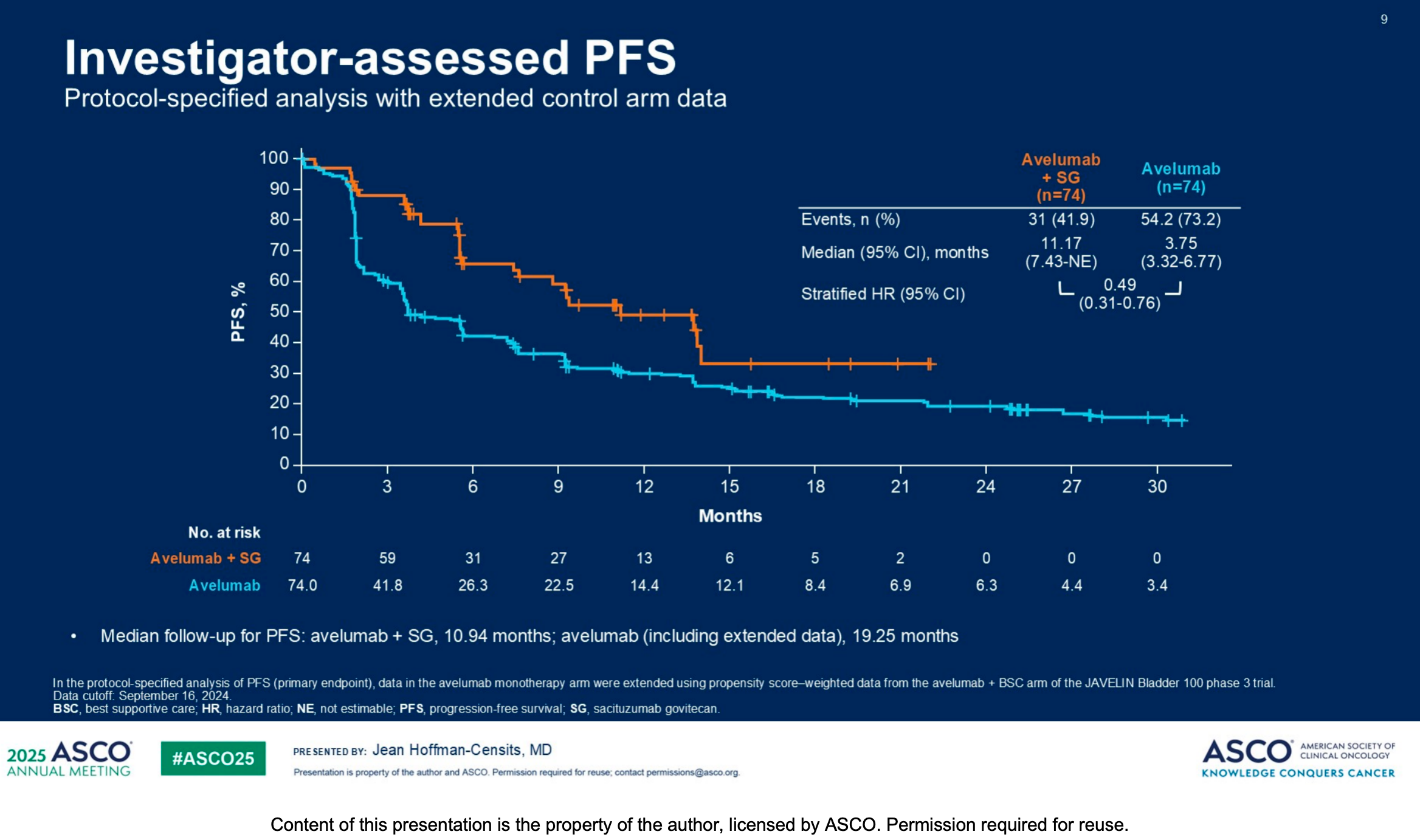
1. Efficacy Data: Overwhelming PFS Advantage
The interim analysis unequivocally demonstrates that PFS was improved with avelumab + SG versus avelumab monotherapy (stratified HR, 0.49 [95% CI, 0.31-0.76]).
Crucially, these PFS improvements were observed across various patient subgroups, indicating the broad clinical applicability of this combination regimen.
OS data were immature at the data cutoff, and longer follow-up is required to further confirm survival benefits.
2. Safety Data: Consistent and Clinically Manageable
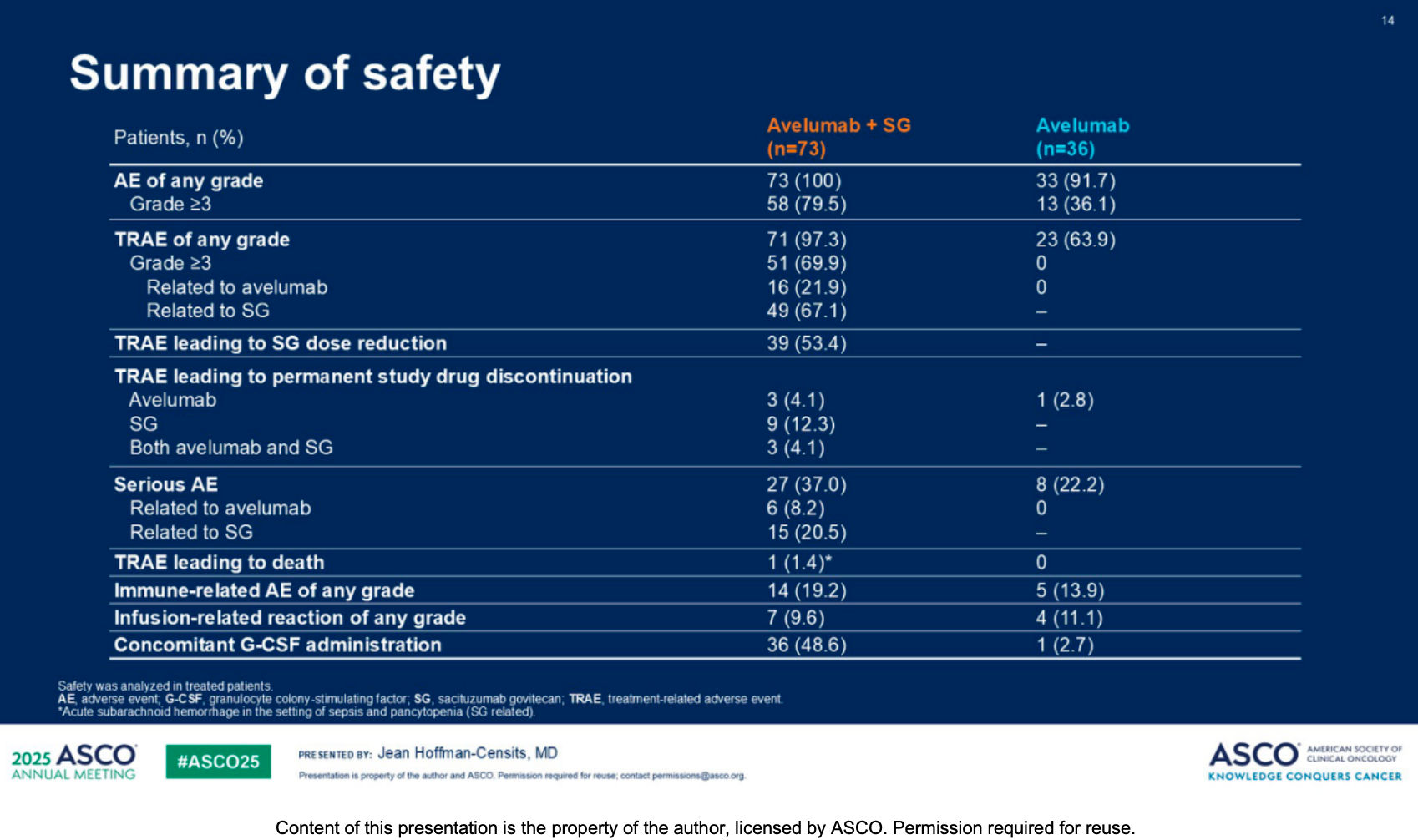
Combination therapies inherently face the challenge of overlapping toxicities. The data show the following treatment-related adverse event (TRAE) profiles for the combination arm:
Avelumab-related AEs: In the combination arm, the most common (≥7.5%) any-grade avelumab-related AEs included fatigue (28.8%), diarrhea (21.9%), asthenia (16.4%), and pruritus (15.1%).
SG-related AEs: The most common any-grade SG-related AEs in the combination arm were alopecia (58.9%), diarrhea (49.3%), neutropenia (47.9%), fatigue (39.7%), and nausea (35.6%).
Overall Assessment: TRAEs were more frequent in the combination arm; however, these events were consistent with the known safety profiles of SG and avelumab, and remain clinically manageable.
V. Conclusions
This interim analysis of the JAVELIN Bladder Medley trial indicates that while the combination of avelumab and sacituzumab govitecan is still an experimental treatment, it demonstrates significant potential as a promising strategy to improve outcomes for patients with advanced UC.
For pharmaceutical companies dedicated to pipeline development and clinical strategy, these data further validate the strategic value of the "PD-(L)1 + ADC" combination in the maintenance treatment phase for solid tumors. A manuscript reporting the full interim analysis results from JAVELIN Bladder Medley has been published in the prestigious medical journal Annals of Oncology, providing the industry with detailed data references.