On April 27, 2026, Intellia Therapeutics announced that it has initiated a rolling Biologics License Application (BLA) submission to the U.S. FDA for its investigational CRISPR gene editing therapy, Lonvoguran Ziclumeran (lonvo-z). The therapy is being developed as a one-time treatment for hereditary angioedema (HAE). The company expects to complete the submission in the second half of 2026, with potential approval anticipated in the first half of 2027.
About Lonvoguran Ziclumeran (lonvo-z)
Lonvoguran Ziclumeran (NTLA-2002) is an innovative gene therapy developed using CRISPR/Cas9 technology. Its defining feature is that it is an in vivo CRISPR gene editing therapy, designed to be administered as a one-time dose in an outpatient setting.
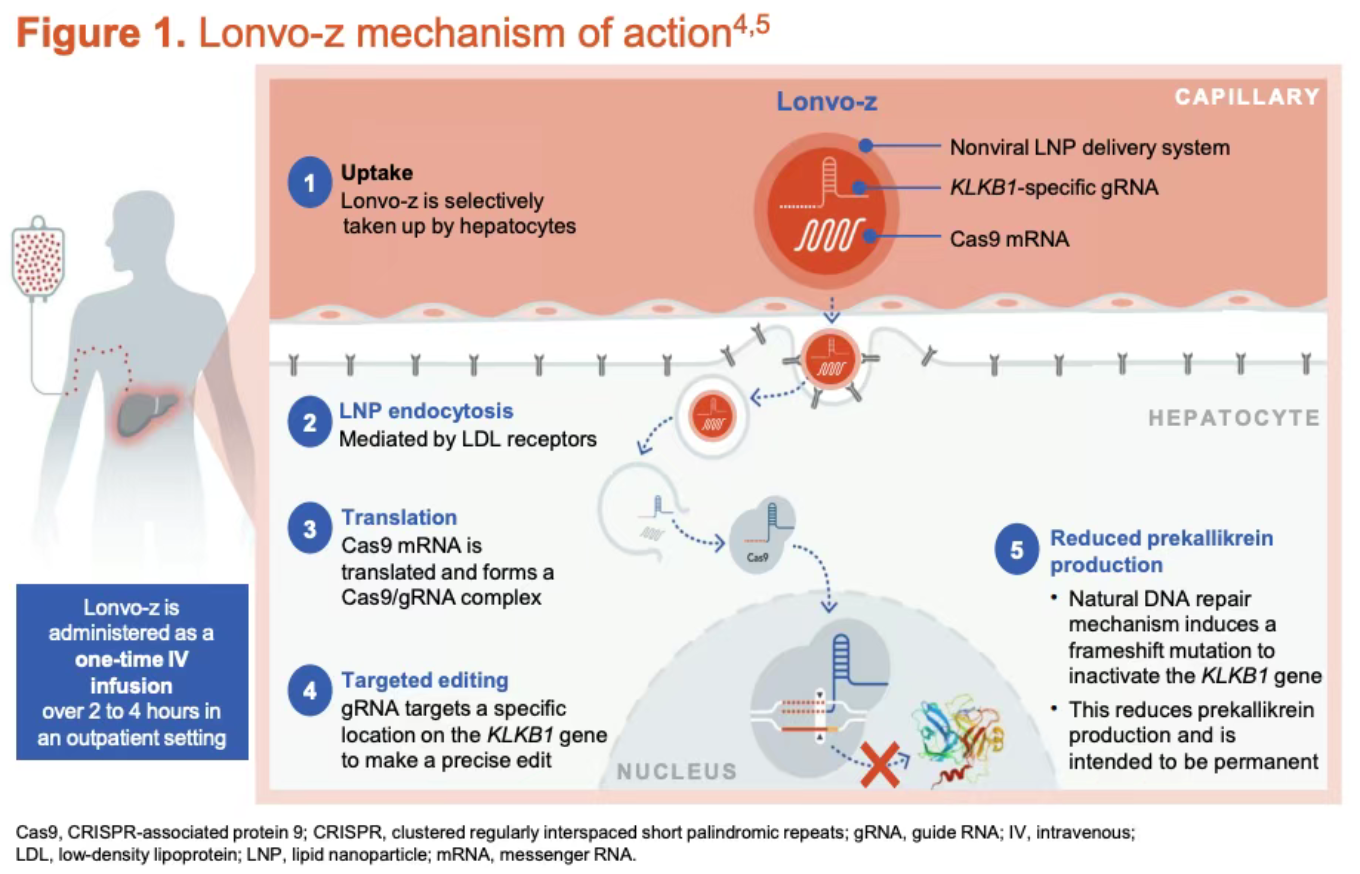
The mechanism of action is highly targeted: through a single injection, the therapy aims to precisely inactivate the KLKB1 gene (kallikrein B1) in patients. Knocking out this gene leads to a permanent reduction in plasma kallikrein and bradykinin levels, thereby fundamentally blocking the pathological pathway responsible for severe swelling attacks.
Given its significant therapeutic potential, lonvo-z has already received multiple major regulatory designations, including:
- Regenerative Medicine Advanced Therapy (RMAT) designation from the U.S. FDA
- Orphan Drug designation
- Priority Medicines (PRIME) designation from the European Medicines Agency (EMA)
- Along with several other global recognitions
Target Disease: Hereditary Angioedema (HAE)
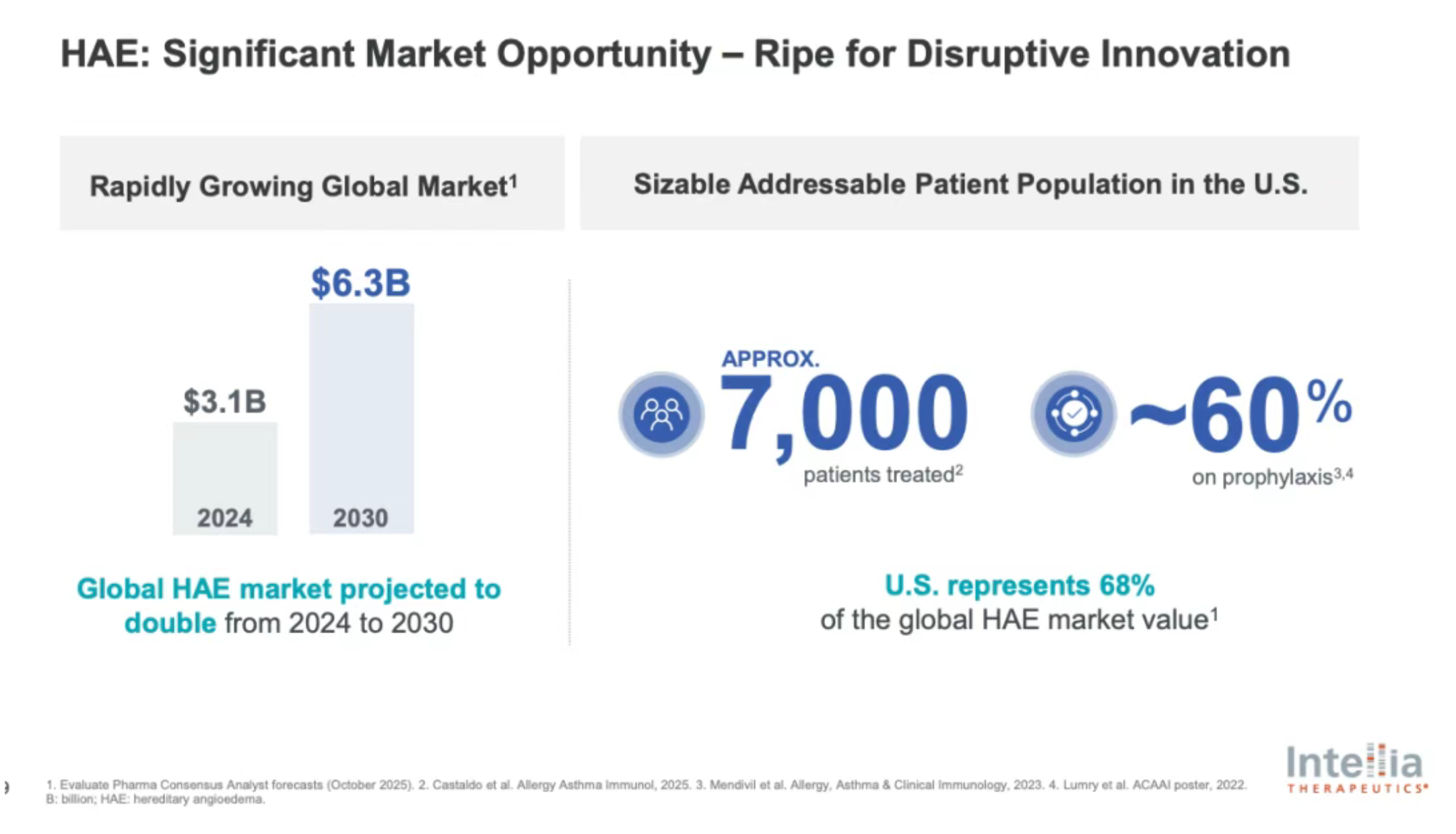
HAE is a rare and potentially life-threatening genetic disorder, with a global prevalence of approximately 1 in 50,000. Patients experience severe, recurrent, and unpredictable inflammatory swelling affecting various organs and tissues.
Current standard of care typically requires lifelong treatment, including:
- Intravenous or subcutaneous injections up to twice weekly
- Or daily oral medications to prevent attacks
Despite such intensive regimens, breakthrough attacks still occur in some patients, imposing substantial physical, psychological, and financial burdens.
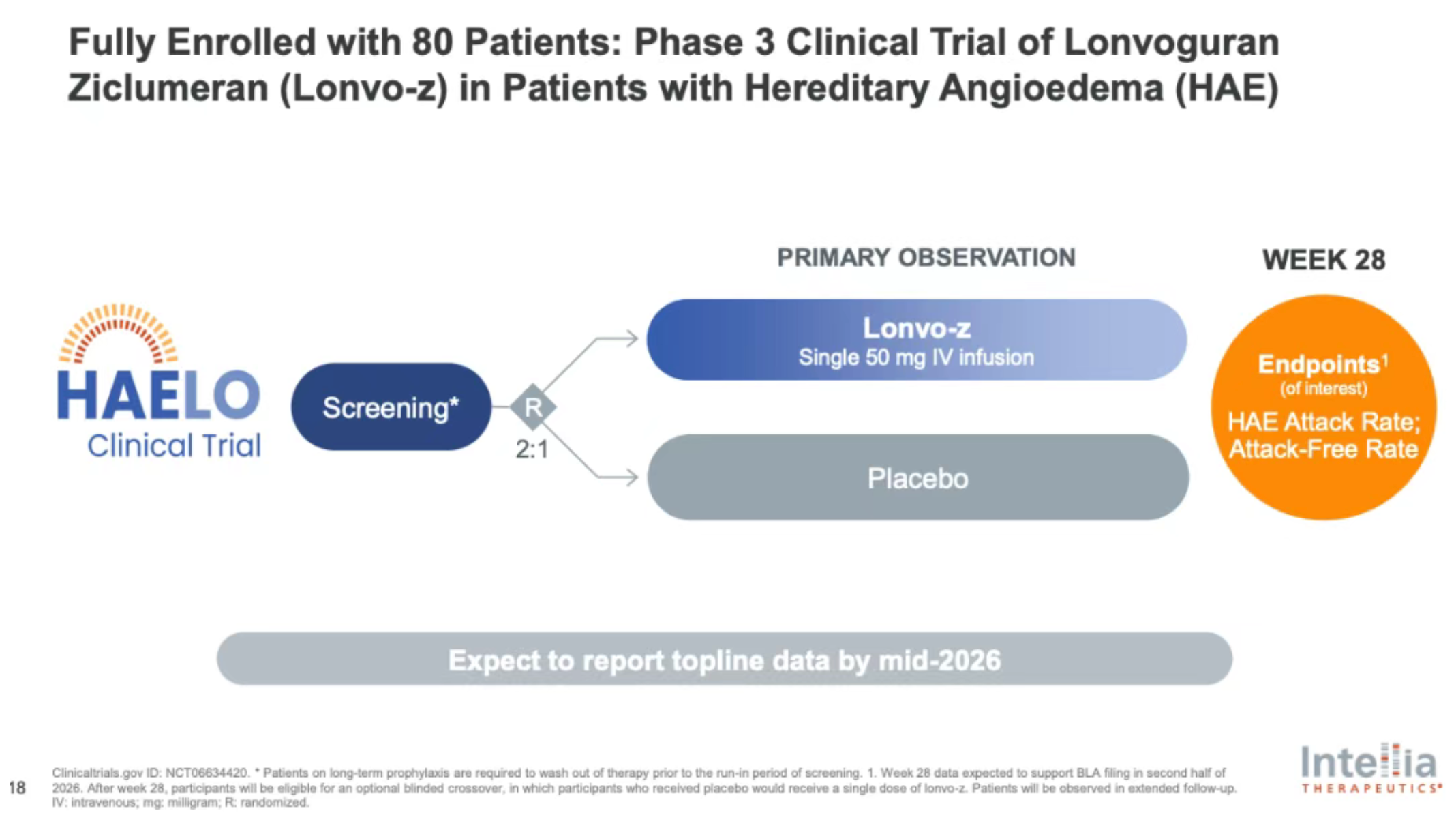
Phase 3 HAELO Trial: Positive Topline Results
On the same day as the BLA announcement, Intellia also released positive topline data from its Phase 3 HAELO trial evaluating lonvo-z in HAE patients.
Key findings include:
- The trial met its primary endpoint and all key secondary endpoints
- During the six-month primary observation period, the vast majority of patients treated with a single dose achieved freedom from attacks
- Patients were able to completely discontinue chronic prophylactic therapies
These striking results suggest that a single injection could enable patients to break free from long-term treatment and recurrent disease burden.
A Milestone in Medicine
1. First In Vivo CRISPR Therapy Approaching Commercialization
While CRISPR therapies have previously been approved, they were ex vivo approaches—requiring extraction of patient cells, laboratory editing, and reinfusion following myeloablative chemotherapy. These procedures are complex, high-risk, and limited to specialized transplant centers.
Lonvo-z ushers in a new era of in vivo treatment, where gene editing tools are delivered directly into the body via a simple outpatient infusion. This breakthrough significantly simplifies the treatment process and broadens accessibility.
2. A Paradigm Shift: From Chronic Management to One-Time Cure
Lonvo-z represents a fundamental shift in rare disease treatment—from lifelong disease management to a potential one-time cure.
As Intellia President and CEO Dr. John Leonard noted, the clinical results reinforce confidence in lonvo-z’s potential to transform the HAE treatment landscape. A single administration could eliminate the need for lifelong preventive therapy and the constant fear of attacks—challenging the traditional “manage but not cure” paradigm.
3. Accelerating Regulatory Pathways for Breakthrough Technologies
Leveraging the FDA’s RMAT designation, Intellia is utilizing a rolling submission process, allowing portions of the application to be reviewed as they are completed—significantly accelerating review timelines.
In addition, the company is participating in the FDA’s pilot program for Chemistry, Manufacturing, and Controls (CMC) development and readiness.
These regulatory “fast-track” mechanisms not only pave the way for lonvo-z’s potential commercialization in the first half of 2027, but also demonstrate strong regulatory support for transformative in vivo gene editing technologies—setting a precedent for future therapies in this space.
Conclusion
Lonvoguran Ziclumeran stands at the forefront of a new generation of genetic medicines. If approved, it could redefine how we treat not only HAE, but potentially a wide range of genetic diseases—transforming gene editing from a complex, niche intervention into a scalable, one-time therapeutic solution.