Key Information
Title
Intracranial Efficacy of Ivonescimab Plus Chemotherapy in Epidermal Factor Receptor (EGFR) Tyrosine Kinase Inhibitor-Resistant, EGFR-Mutated Non-Small Cell Lung Cancer in the HARMONI Study
Drug Name
Ivonescimab Pemetrexed Carboplatin
Mechanism of Action
Bispecific antibody against programmed cell death protein 1 (PD-1) and vascular endothelial growth factor (VEGF)
Indication
EGFR-mutated non-squamous non-small cell lung cancer (NSCLC) progressed on a third-generation EGFR-TKI
Phase
Phase 3
Clinical Trial ID
NCT06396065NCT05184712
Objective
To explore intracranial disease control by assessing intracranial progression-free survival (PFS) and cumulative incidence of intracranial progression by baseline brain metastases status in patients with EGFR-mutated NSCLC who progressed on a third-generation EGFR-TKI.
Original Interpretation
BACKGROUND
• Ivonescimab is an investigational bispecific antibody against programmed cell death protein 1 (PD-1) and vascular endothelial growth factor (VEGF) 11
- In patients with EGFR-tyrosine kinase inhibitor (TKI)–pretreated, advanced or metastatic EGFR-mutated non–small cell lung cancer (NSCLC), ivonescimab + chemotherapy significantly prolonged progression-free survival (PFS) and overall survival (OS) compared with chemotherapy alone in the phase 3 HARMONi-A trial (NCT05184712) conducted in China 2,32,3
> Based on this study, ivonescimab + chemotherapy was approved in China for the second-line treatment of EGFR-mutated NSCLC 44
- HARMONi is a global, randomized, double-blind, phase 3 trial evaluating the efficacy and safety of ivonescimab + chemotherapy versus placebo + chemotherapy in patients with EGFR-mutated non-squamous NSCLC whose disease has progressed on a third-generation EGFR-TKI 5,65,6
In the primary analysis of HARMONi, PFS as assessed by independent radiology review committee (IRRC) was longer with ivonescimab + chemotherapy compared with placebo + chemotherapy (hazard ratio [HR], 0.52; 95% confidence interval [CI], 0.41-0.66) 66
» The PFS HR was 0.34 (95% CI, 0.20-0.57) in patients with brain metastases at baseline and 0.59 (95% CI, 0.45-0.77) in patients without brain metastases at baseline 66
> Grade ≥3 treatment-related adverse events (TRAEs) were reported in 50.0% and 42.2% of patients and serious TRAEs in 28.0% and 15.1% of patients in the ivonescimab + chemotherapy and placebo + chemotherapy arms, respectively 66
» Rates of TRAEs leading to study drug discontinuation (7.3% vs 5.0%) and TRAEs leading to death (1.8% vs 2.3%) were comparable with ivonescimab + chemotherapy versus placebo + chemotherapy 66
- To explore intracranial disease control, we assessed intracranial PFS and cumulative incidence of intracranial progression by baseline brain metastases status
Intracranial endpoints were evaluated in prespecified exploratory analyses of HARMONi and were not part of the primary multiplicity-controlled testing hierarchy
METHODS
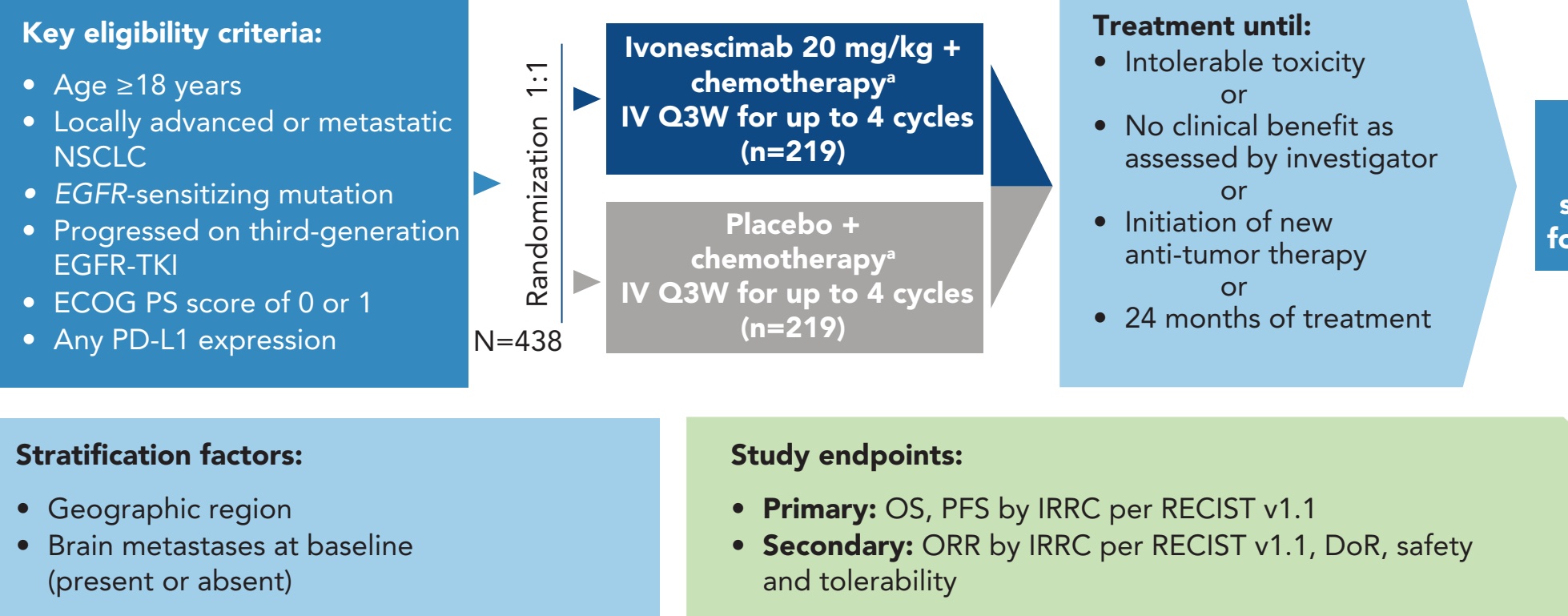
• In HARMONi (NCT06396065), eligible patients were randomly assigned (1:1) to receive ivonescimab (20 mg/kg) or placebo with pemetrexed and carboplatin intravenously every 3 weeks for 4 cycles, followed by maintenance therapy with ivonescimab or placebo + pemetrexed for up to 24 months of treatment (Figure 1) 5,65,6 Patients with symptomatic metastases of the central nervous system were excluded
Patients with symptomatic metastases of the central nervous system were excluded
Figure 1. Trial Design
[Interpretation]
The image illustrates the clinical trial design for the HARMONi study (NCT06396065), a Phase 3 trial evaluating the efficacy and safety of ivonescimab in combination with chemotherapy for patients with specific types of non-small cell lung cancer (NSCLC).
Figure 1: HARMONi Trial Design
This flowchart outlines the structured approach of the study, from patient selection to treatment and endpoint evaluation:
-
Key Eligibility Criteria:
- Adults (age ≥18 years) with locally advanced or metastatic NSCLC.
- Tumors must harbor an EGFR-sensitizing mutation.
- Patients must have progressed on a third-generation EGFR tyrosine kinase inhibitor (TKI) (e.g., osimertinib).
- An ECOG performance status (PS) of 0 or 1 is required.
- Enrollment is open regardless of PD-L1 expression levels.
- Adults (age ≥18 years) with locally advanced or metastatic NSCLC.
- Tumors must harbor an EGFR-sensitizing mutation.
- Patients must have progressed on a third-generation EGFR tyrosine kinase inhibitor (TKI) (e.g., osimertinib).
- An ECOG performance status (PS) of 0 or 1 is required.
- Enrollment is open regardless of PD-L1 expression levels.
-
Study Design and Randomization:
- A total of 438 patients (N=438) were randomized in a 1:1 ratio into two treatment arms:
- Ivonescimab Arm (n=219): Ivonescimab (20 mg/kg) plus chemotherapy (pemetrexed and carboplatin) administered intravenously every 3 weeks (Q3W) for up to 4 cycles.
- Placebo Arm (n=219): Placebo plus chemotherapy administered intravenously Q3W for up to 4 cycles.
- Following the initial 4 cycles, patients continued maintenance therapy with either ivonescimab or placebo plus pemetrexed.
- A total of 438 patients (N=438) were randomized in a 1:1 ratio into two treatment arms:
- A total of 438 patients (N=438) were randomized in a 1:1 ratio into two treatment arms:
- Ivonescimab Arm (n=219): Ivonescimab (20 mg/kg) plus chemotherapy (pemetrexed and carboplatin) administered intravenously every 3 weeks (Q3W) for up to 4 cycles.
- Placebo Arm (n=219): Placebo plus chemotherapy administered intravenously Q3W for up to 4 cycles.
- Ivonescimab Arm (n=219): Ivonescimab (20 mg/kg) plus chemotherapy (pemetrexed and carboplatin) administered intravenously every 3 weeks (Q3W) for up to 4 cycles.
- Placebo Arm (n=219): Placebo plus chemotherapy administered intravenously Q3W for up to 4 cycles.
- Following the initial 4 cycles, patients continued maintenance therapy with either ivonescimab or placebo plus pemetrexed.
-
Treatment Duration:
- Treatment continued until the occurrence of intolerable toxicity, loss of clinical benefit (as assessed by the investigator), initiation of a new anti-tumor therapy, or completion of 24 months of treatment.
- Treatment continued until the occurrence of intolerable toxicity, loss of clinical benefit (as assessed by the investigator), initiation of a new anti-tumor therapy, or completion of 24 months of treatment.
-
Stratification Factors:
- Randomization was stratified by geographic region and the presence or absence of brain metastases at baseline. The inclusion of brain metastases as a stratification factor underscores the study's specific interest in intracranial efficacy, as noted in the poster title.
- Randomization was stratified by geographic region and the presence or absence of brain metastases at baseline. The inclusion of brain metastases as a stratification factor underscores the study's specific interest in intracranial efficacy, as noted in the poster title.
-
Study Endpoints:
- Primary Endpoints: Overall Survival (OS) and Progression-Free Survival (PFS) as assessed by an Independent Radiology Review Committee (IRRC) per RECIST v1.1.
- Secondary Endpoints: Objective Response Rate (ORR), Duration of Response (DoR), and safety/tolerability profile.
- Primary Endpoints: Overall Survival (OS) and Progression-Free Survival (PFS) as assessed by an Independent Radiology Review Committee (IRRC) per RECIST v1.1.
- Secondary Endpoints: Objective Response Rate (ORR), Duration of Response (DoR), and safety/tolerability profile.
Overall, this design aims to provide high-level clinical evidence on whether adding ivonescimab—a first-in-class bispecific antibody targeting both PD-1 and VEGF—to standard chemotherapy can improve outcomes for EGFR-mutated NSCLC patients who have exhausted standard TKI options.
Study endpoints:
• Geographic region • Brain metastases at baseline (present or absent)
- Primary: OS, PFS by IRRC per RECIST v1.1 - Secondary: ORR by IRRC per RECIST v1.1, DoR, safety and tolerability
AUC, area under the concentration-time curve; DoR, duration of response; ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR-TKI, epidermal growth factor receptor-tyrosine kinase inhibitor; IRRC, independent radiology review committee; NSCLC, non–small cell lung cancer; ORR, objective response rate; OS, overall survival; PD-L1, programmed cell death ligand-1; PFS, progression-free survival; Q3W, every 3 weeks; RECIST v1.1, Response Evaluation Criteria in Solid Tumors, version 1.1. aaChemotherapy was pemetrexed 500 mg/m 22 and carboplatin AUC 5 mg/mL/min. Carboplatin was discontinued after an induction phase of 4 cycles.
- This subgroup analysis evaluated intracranial PFS (as assessed by IRRC based on Response Evaluation Criteria in Solid Tumors, version 1.1 [RECIST v1.1]) and cumulative incidence probability of intracranial progression, defined as occurrence of a new brain lesion or progression of an existing brain lesion, in patients with and without brain metastases at baseline The probability of a first event being an intracranial progression, non-intracranial progression, or death was estimated by cumulative incidences using a competing risks approach in patients
At screening, an enhanced scan of the brain by computed tomography or magnetic resonance imaging (MRI) was performed (if not contraindicated) to evaluate for brain metastases; if suspected, an MRI scan of the brain was required to confirm or reject the diagnosis
During the study, if brain metastases were present at baseline, brain imaging was performed with repeat MRI per the tumor imaging schedule: every 6 weeks ( ±7±7 days) for 54 weeks after the first dose of study treatment, then every 12 weeks ( ±7±7 days)
> If clinically indicated, the investigators were allowed to perform additional scans or more frequent evaluations
Patient Characteristics
• Of the 438 patients who were randomized to receive ivonescimab + chemotherapy (n=219) or placebo + chemotherapy (n=219), 54 (24.7%) in each arm had brain metastases at baseline (Table 1)
RESULTS
The demographics and baseline disease characteristics were generally balanced between treatment arms for patients with and without brain metastases
Intracranial PFS
- At the data cutoff of April 12, 2025, median intracranial PFS was longer in the ivonescimab + chemotherapy arm versus the placebo + chemotherapy arm in patients with and without brain metastases (Figure 2)
Cumulative Incidence Probability for Intracranial Progression
- The cumulative incidence probability (95% CI) for intracranial progression at 6 months and 12 months was significantly lower with ivonescimab + chemotherapy versus placebo + chemotherapy in both subgroups (Figure 3)
> For patients without brain metastases, the benefit continued for more than 27 months
Table 1. Baseline Characteristics
Patients with brain metastases Patients without brain metastases Ivo + chemo Placebo + chemo Ivo + chemo Placebo + chemo Characteristic n=54 n=54 n=165 n=165 Age, median (range), years 63 (40.0-84.0) 58 (37.0-84.0) 62 (32.0-82.0) 62 (36.0-81.0) Age ≥65 years, n (%) 21 (38.9) 19 (35.2) 62 (37.6) 69 (41.8) Female sex, n (%) 33 (61.1) 30 (55.6) 97 (58.8) 97 (58.8) Region, n (%) Asia 29 (53.7) 31 (57.4) 107 (64.8) 106 (64.2) Europe 10 (18.5) 9 (16.7) 30 (18.2) 23 (13.9) North America 15 (27.8) 14 (25.9) 28 (17.0) 36 (21.8) Race, n (%) American Indian or Alaska Native 0 0 0 1 (0.6) Asian 34 (63.0) 36 (66.7) 119 (72.1) 117 (70.9) Black or African American 2 (3.7) 0 0 3 (1.8) White 13 (24.1) 16 (29.6) 38 (23.0) 38 (23.0) Other aa 5 (9.3) 2 (3.7) 8 (4.8) 6 (3.6) Never-smoker, n (%) 39 (72.2) 41 (75.9) 104 (63.0) 114 (69.1) Liver metastasis, n (%) 10 (18.5) 10 (18.5) 22 (13.3) 13 (7.9) Prior lines of systemic anti-cancer therapy, median (range) 1 (1.0-3.0) 1 (1.0-3.0) 1 (1.0-4.0) 1 (1.0-4.0)
[Interpretation]
The baseline characteristics presented in Table 1 from the HARMONI study provide a critical foundation for evaluating the intracranial efficacy of ivonescimab (a PD-1/VEGF bispecific antibody) plus chemotherapy.
1. Balanced and Comparable Study Arms
The most significant finding from Table 1 is the high degree of balance between the ivonescimab and placebo arms within both subgroups (patients with and without brain metastases). Key metrics—including median age (58–63 years), sex distribution, and geographical region—are nearly identical across arms. This balance is crucial for a professional interpretation, as it ensures that the reported improvements in intracranial progression-free survival (iPFS) and reduced cumulative incidence of progression are attributable to the study drug rather than baseline discrepancies.
2. Representative EGFR-Mutated NSCLC Population
The demographic profile aligns perfectly with the typical epidemiology of EGFR-mutated non-small cell lung cancer (NSCLC):
- Sex and Smoking Status: There is a high prevalence of females (55.6%–61.1%) and never-smokers (63.0%–75.9%). These are hallmark characteristics of the EGFR-driven disease phenotype.
- Ethnicity and Region: The study is global but has a strong Asian representation (~63%–72% Asian race). Given that EGFR mutations are significantly more prevalent in Asian populations (approx. 40–50% compared to 10–15% in Western populations), this demographic makeup ensures the findings are highly relevant to the primary target patient group.
3. High-Risk Clinical Features
The table highlights a population with a high disease burden and significant resistance:
- TKI Resistance: All patients had previously received systemic anti-cancer therapy (median of 1 prior line), confirming this as an EGFR TKI-resistant cohort.
- Liver Metastases: Approximately 13.3% to 18.5% of patients had liver metastases at baseline. Liver involvement is traditionally a poor prognostic factor in NSCLC and often correlates with lower response rates to immunotherapy, making the efficacy of the ivonescimab combination in this group particularly noteworthy.
4. Strategic Subgrouping for Intracranial Analysis
The division into patients "With" (n=108) and "Without" (n=330) brain metastases is a critical signal. Patients with EGFR mutations are at high risk for central nervous system (CNS) progression. By maintaining balanced cohorts in these subgroups, the study provides robust evidence for ivonescimab's ability to cross or impact the blood-brain barrier—a common failure point for many systemic therapies.
Conclusion
The baseline data confirms that the HARMONI study targeted a well-defined, high-risk, TKI-resistant population. The consistency across arms validates the subsequent efficacy findings: the prolonged intracranial PFS and the "benefit continuing for more than 27 months" in patients without brain metastases can be viewed with high confidence as a true therapeutic signal of the ivonescimab-chemotherapy combination.
Figure 2. Intracranial PFS in Patients (A) With and (B) Without Brain Metastases aa
aaOther includes unknown and not permitted per regulation.

[Interpretation]
This image displays a Kaplan-Meier plot and a summary table representing Figure 2B from a medical poster. It details the Intracranial Progression-Free Survival (iPFS) for a specific subgroup of patients (likely those with baseline brain metastases, given the sample size of n=54 per arm) in the HARMONI study.
Sub-figure: Intracranial PFS Kaplan-Meier Plot (Panel B)
The chart compares the efficacy of Ivonescimab + chemotherapy (blue line) against Placebo + chemotherapy (grey line) in preventing intracranial disease progression or death.
Data Summary Table
Metric Ivo + chemo (n=54) Placebo + chemo (n=54) No. of events 34 39 Median PFS (95% CI), months 10.1 (8.4-13.4) 6.5 (5.1-9.2) HR (95% CI) - 0.53 (0.33-0.84); P=0.0068
Professional Interpretation
- Significant Clinical Benefit: The combination of Ivonescimab (a PD-1/VEGF bi-specific antibody) and chemotherapy demonstrated a statistically significant improvement in intracranial PFS compared to chemotherapy alone.
- Survival Metrics: The median intracranial PFS was extended by 3.6 months (10.1 months for the ivonescimab group vs. 6.5 months for the placebo group).
- Risk Reduction: The Hazard Ratio (HR) of 0.53 indicates that patients receiving the ivonescimab combination had a 47% reduction in the risk of intracranial progression or death compared to the control group.
- Curve Analysis: The Kaplan-Meier curves begin to separate early (at the first assessment point around 3 months) and maintain a consistent gap throughout the follow-up period, suggesting early and sustained intracranial activity. The p-value of 0.0068 confirms that this observed benefit is highly unlikely to be due to chance.
- Context: In the setting of EGFR-mutated NSCLC that is resistant to TKIs, managing brain metastases is a critical challenge. These data suggest that ivonescimab's dual inhibition of PD-1 and VEGF may enhance CNS disease control, potentially due to the role of VEGF inhibition in normalizing tumor vasculature and reducing peritumoral edema in the brain.
No. patients at risk (censored)
Ivonescimab + chemotherapy 54 (0) 49 (3) 37 (7) 22 (12) 14 (15) 9 (16) 7 (17) 4 (18) 3 (19) 1 (19) 1 (19) 0 (20) Placebo + chemotherapy 54 (0) 39 (1) 20 (10) 12 (12) 7 (14) 5 (15) 1 (15) 1 (15) 1 (15) 1 (15) 0 (15)
Chemo, chemotherapy; HR, hazard ratio; ivo, ivonescimab; PFS, progression-free survival. aaThese analyses were descriptive.

[Interpretation]
The provided image displays a Kaplan-Meier plot for Intracranial Progression-Free Survival (iPFS) from the HARMONI study. This study investigates the efficacy of Ivonescimab (a PD-1/VEGF bispecific antibody) plus chemotherapy versus placebo plus chemotherapy in patients with EGFR-mutated non-small cell lung cancer (NSCLC) who are resistant to EGFR tyrosine kinase inhibitors (TKIs).
Data Summary Table
The following table reproduces the data presented in the inset of the figure:
Metric Ivo + chemo (n=165) Placebo + chemo (n=165) No. of events 96 112 Median PFS (95% CI), months 15.7 (13.4-17.1) 11.6 (10.1-13.6) HR (95% CI) - 0.72 (0.55-0.94) P-value - P=0.0172
Professional Interpretation
The chart demonstrates a statistically significant and clinically meaningful improvement in intracranial progression-free survival for patients treated with Ivonescimab plus chemotherapy compared to those receiving placebo plus chemotherapy.
1. Prolonged Median Survival: The median iPFS was extended by 4.1 months in the Ivonescimab group (15.7 months vs. 11.6 months). This is a critical finding for this patient population, as brain metastases and intracranial progression are significant causes of morbidity and mortality in EGFR-mutated NSCLC.
2. Risk Reduction: The Hazard Ratio (HR) of 0.72 indicates that the addition of Ivonescimab to chemotherapy reduces the risk of intracranial disease progression or death by 28% (95% CI: 0.55-0.94). The upper bound of the confidence interval being below 1.0, combined with a p-value of 0.0172, confirms the statistical significance of this benefit.
3. Kinetic Analysis of the Curve: The survival curves begin to separate early (around month 3) and remain separated throughout the 36-month observation period. This sustained separation suggests that the therapeutic benefit of the bispecific antibody (targeting both PD-1 and VEGF) provides durable intracranial control.
4. Clinical Context: In the context of EGFR-TKI resistance, finding effective therapies that can penetrate or act within the central nervous system (CNS) is challenging. The efficacy shown here suggests that the combination of Ivonescimab and chemotherapy may enhance CNS disease control, possibly through the synergistic effects of VEGF inhibition (which can normalize tumor vasculature and reduce edema) and immune checkpoint inhibition.
No. patients at risk (censored)
Figure 3. Cumulative Incidence Probability for Intracranial Progression in Patients (A) With and (B) Without Brain Metastases
Ivonescimab + chemotherapy 165 (0) 153 (6) 124 (17) 84 (40) 62 (48) 51 (50) 34 (54) 24 (57) 16 (59) 10 (61) 6 (63) 3 (66) 0 (69) Placebo + chemotherapy 165 (0) 144 (9) 108 (21) 72 (39) 54 (43) 37 (47) 29 (49) 21 (50) 16 (50) 8 (51) 3 (53) 2 (53) 0 (53)

[Interpretation]
This image displays a cumulative incidence curve representing the probability of intracranial progression over time for patients in the HARMONI study. Based on the provided context, this specific chart corresponds to Figure 3A, focusing on patients with brain metastases at baseline (n=54 per treatment arm).
Cumulative Incidence Probability for Intracranial Progression
The chart compares the cumulative incidence of intracranial progression between two treatment groups:
- Ivonescimab + chemotherapy (dark blue line)
- Placebo + chemotherapy (grey line)
Key Observations:
- Significant Reduction in Progression: There is a clear and widening separation between the two curves starting very early in the treatment (around 2 months). The Ivonescimab + chemotherapy arm demonstrates a substantially lower cumulative incidence of intracranial progression compared to the placebo arm.
- Early Efficacy: By month 3, the placebo group shows a sharp increase in cumulative incidence (reaching ~0.2), while the Ivonescimab group remains nearly at zero.
- Long-term Benefit: The benefit is sustained over time. While the placebo arm reaches a cumulative incidence of nearly 0.4 by month 12, the Ivonescimab arm remains significantly lower, staying below 0.2 even beyond 15 months.
- Statistical Significance: The P-value of 0.0007 indicates that the reduction in the risk of intracranial progression with Ivonescimab + chemotherapy is highly statistically significant.
Summary Data Table
The following table, embedded within the chart, provides specific incidence rates and confidence intervals:
Timepoint Ivo + chemo (n=54) Placebo + chemo (n=54) 6 months (95% CI) 0.061 (0.015-0.152) 0.304 (0.185-0.432) 12 months (95% CI) 0.104 (0.037-0.210) 0.357 (0.224-0.493) P-value 0.0007
Professional Analysis of Table Data: The numerical data reinforces the visual trends of the curve. At 6 months, patients receiving Ivonescimab + chemotherapy had an approximately 80% lower relative incidence of intracranial progression compared to the placebo group (6.1% vs 30.4%). By 12 months, the disparity remains stark, with the Ivonescimab group showing a 10.4% incidence versus 35.7% in the placebo group. These results strongly suggest that the addition of Ivonescimab to chemotherapy provides superior intracranial disease control in EGFR-TKI resistant, EGFR-mutated NSCLC patients with existing brain metastases.
Cumulative no. of events Ivonescimab + chemotherapy 0 1 3 5 5 6 7 7 7 7 7 7 Placebo + chemotherapy 0 11 16 18 18 19
[Interpretation]
This data from the HARMONI study evaluates the intracranial efficacy of ivonescimab (a PD-1/VEGF bispecific antibody) combined with chemotherapy in patients with EGFR-mutated non-small cell lung cancer (NSCLC) who have failed prior TKI therapy.
The provided table tracks the cumulative number of intracranial progression events over time. Based on this data, several core findings and clinical signals emerge:
1. Significant Reduction in Intracranial Progression
The most critical signal is the stark difference in the total number of intracranial events between the two arms.
- Ivonescimab + Chemotherapy: Recorded only 7 cumulative events by the end of the observation period.
- Placebo + Chemotherapy: Recorded 19 cumulative events before the data tracking concluded.
This represents a substantial reduction (nearly 63%) in the absolute number of patients experiencing intracranial progression when ivonescimab is added to the treatment regimen.
2. Early and Sustained CNS Protection
The temporal trend of these events reveals a "protective" effect that begins almost immediately:
- Rapid Progression in Placebo Arm: In the placebo arm, 11 events occurred by the first follow-up interval, surging to 16 by the second. This suggests that chemotherapy alone provides poor control over CNS disease in this resistant population.
- Delayed/Controlled Progression in Ivonescimab Arm: In contrast, the ivonescimab arm showed only 1 event at the first interval and 3 at the second. The progression curve is significantly flatter, indicating that ivonescimab effectively delays the onset of brain metastases or the progression of existing ones.
3. Therapeutic Synergy (PD-1/VEGF Inhibition)
From a professional perspective, these results underscore the importance of VEGF inhibition in managing CNS disease. VEGF is a key driver of vascular permeability and edema in brain metastases. By combining PD-1 blockade with VEGF inhibition, ivonescimab likely improves the "immunosupportive" nature of the tumor microenvironment in the brain, potentially enhancing the delivery and efficacy of both the immunotherapy and chemotherapy components.
4. Clinical Implications for EGFR-Mutated NSCLC
Patients with EGFR-mutated NSCLC have a high lifetime risk of developing brain metastases. The "at risk" data (showing 165 patients in each arm for Figure 3B) suggests this study was sufficiently powered to observe these trends. The ability of ivonescimab to maintain a plateau of only 7 events from the seventh time point onward suggests long-term intracranial stability for responders.
5. Safety Context
The surrounding text indicates that this intracranial efficacy does not come at the cost of significantly increased toxicity. Grade $\geq$3 treatment-related adverse events (TRAEs) and immune-related TRAEs were similar between patients with and without brain metastases. This confirms that the CNS-specific benefits of ivonescimab are achievable with a manageable safety profile.
Conclusion
The data demonstrates that ivonescimab plus chemotherapy provides superior intracranial disease control compared to chemotherapy alone. The combination significantly reduces the incidence and delays the onset of intracranial progression, addressing a critical unmet need for patients with EGFR-mutated NSCLC who have progressed on TKI therapy.
Chemo, chemotherapy; HR, hazard ratio; ivo, ivonescimab.

[Interpretation]
This image displays a cumulative incidence curve from the HARMONI study, specifically representing Figure 3B: Cumulative Incidence Probability for Intracranial Progression in Patients Without Brain Metastases.
Data Summary Table
The table inset within the figure provides the statistical breakdown of the incidence of intracranial progression at key time points:
Time Point Ivonescimab + chemo (n=165) Placebo + chemo (n=165) 6 months (95% CI) 0.025 (0.008–0.059) 0.081 (0.045–0.130) 12 months (95% CI) 0.032 (0.012–0.068) 0.095 (0.055–0.147) P-value 0.0136
Professional Interpretation
This chart evaluates the ability of ivonescimab (a PD-1/VEGF bispecific antibody) combined with chemotherapy to prevent new intracranial progression in patients with EGFR-mutated, TKI-resistant non-small cell lung cancer (NSCLC) who did not have brain metastases at the start of the study.
- Primary Finding: The combination of ivonescimab and chemotherapy significantly reduced the risk of developing brain metastases compared to the placebo plus chemotherapy arm. The cumulative incidence curves separate early (around 2 months) and remain widely separated throughout the 36-month observation period.
- Quantitative Signal: At 12 months, the cumulative incidence of intracranial progression was only 3.2% in the ivonescimab arm, compared to 9.5% in the placebo arm. This represents a nearly 3-fold reduction in the probability of the cancer spreading to the brain within the first year of treatment.
- Statistical Significance: The difference between the two treatment groups is statistically significant, with a p-value of 0.0136.
- Clinical Relevance: For patients with EGFR-mutated NSCLC, the brain is a frequent site of progression after failure of TKI therapy (like osimertinib). These data suggest that ivonescimab, by potentially enhancing drug delivery or immune surveillance through its dual VEGF and PD-1 inhibition, provides superior intracranial protection, which is a critical clinical benefit for long-term patient management and quality of life.
Time (months)
Cumulative no. of events Ivonescimab + chemotherapy 0 2 4 5 5 5 5 5 5 5 5 5 5 5 Placebo + chemotherapy 0 7 13 15 15 15 16 16 16 16 16 16
[Interpretation]
Based on the provided data from the HARMONI study, the following professional interpretation focuses on the intracranial efficacy and safety of Ivonescimab plus chemotherapy in patients with EGFR-mutated NSCLC.
Core Findings: Intracranial Efficacy
The primary metric provided in the table is the cumulative number of intracranial progression events over time. The data reveals a significant clinical signal:
- Substantial Reduction in CNS Progression: The Ivonescimab + chemotherapy arm demonstrated a dramatic reduction in intracranial events compared to the placebo arm. Total events were capped at 5 for the Ivonescimab group, compared to 16 in the placebo group—representing a nearly 70% lower incidence of brain progression.
- Early and Sustained Stabilization: In the Ivonescimab arm, the cumulative events plateaued extremely early (at month 3), with no new intracranial progression events recorded from month 3 through month 13. Conversely, the placebo arm showed a rapid early surge (7 events by the first milestone) and continued to accrue events until stabilizing much later at a higher threshold.
- Superior Durability: The "Number of Patients at Risk" data indicates that while both groups saw attrition, the Ivonescimab group maintained a higher proportion of patients free from intracranial progression for a longer duration. This suggests that Ivonescimab, a bispecific antibody targeting both PD-1 and VEGF, may effectively modulate the tumor microenvironment within the central nervous system (CNS).
Clinical Context and Critical Signals
- Targeting a High-Need Population: EGFR-mutated NSCLC patients who have failed TKI therapy are at high risk for CNS relapse. The ability of the Ivonescimab regimen to "flatline" the cumulative event rate early in treatment suggests a potent protective effect against brain metastasis progression, which is often a major driver of morbidity and mortality in this population.
- The Role of VEGF Inhibition: The efficacy likely stems from the synergistic effect of VEGF inhibition (which reduces peritumor edema and improves drug delivery) and PD-1 blockade. This is particularly relevant in the CNS, where VEGF plays a critical role in blood-brain barrier permeability.
Safety and Tolerability
The surrounding text confirms that this intracranial efficacy does not come at the expense of disproportionate toxicity:
- Subgroup Consistency: Frequencies of grade ≥3 Treatment-Emergent Adverse Events (TRAEs) and immune-related TRAEs were similar between patients with and without brain metastases.
- Manageable Profile: Serious immune-related events (like grade ≥3 pneumonitis or transaminase elevations) were rare, occurring in only 1–2 patients per subgroup. This indicates that the addition of Ivonescimab to chemotherapy is a tolerable strategy for patients with CNS involvement.
Conclusion
The HARMONI study data provides a compelling professional rationale for the use of Ivonescimab plus chemotherapy in TKI-resistant EGFR-mutated NSCLC. The critical signal is the rapid and sustained prevention of intracranial progression, establishing a high bar for CNS control in a difficult-to-treat patient population.
Safety
- The frequencies of grade 3 or higher TRAEs were generally similar in subgroups of patients with and without brain metastases for each treatment arm (Table 2)
• Immune-related TRAEs of any grade occurred at similar frequencies in patients with and without brain metastases within each treatment arm (Table 2)
> In the ivonescimab + chemotherapy arm, grade ≥≥3 immune-related TRAEs that occurred in >1 patient were increased alanine aminotransferase (1 patient in each subgroup), drug eruption (2 patients without brain metastases), and increased lipase (2 patients without brain metastases)
» Grade ≥≥3 interstitial lung toxicity (including pneumonitis, interstitial lung disease, and immune-mediated lung disease) occurred in 1 patient with and 2 patients without brain metastases, respectively in the ivonescimab + chemotherapy arm
• VEGF inhibition-associated TRAEs of any grade also occurred at similar frequencies in patients with and without brain metastases within each treatment arm (Table 2)
> For patients in the ivonescimab + chemotherapy arm, grade ≥≥3 VEGF-related TRAEs by classification that occurred in >1 patient were hypertension (2 patients with and 6 patients without brain metastases), proteinuria (2 patients without brain metastases), hemorrhage (2 patients without brain metastases), congestive heart failure (2 patients without brain metastases), and venous thrombotic events (4 patients without brain metastases)
• The frequencies of TRAEs in patients in the ivonescimab + chemotherapy arm were generally consistent across subgroups (Table 3)
Table 2. Summary of TRAEs
Patients with brain metastases Patients without brain metastases Characteristic, n (%) Ivo + chemo Placebo + chemo Ivo + chemo Placebo + chemo n=54 n=54 n=164 n=164 TRAEs (all grades) 48 (88.9) 49 (90.7) 159 (97.0) 154 (93.9) Grade ≥3 26 (48.1) 23 (42.6) 83 (50.6) 69 (42.1) Serious TRAEs 11 (20.4) 7 (13.0) 50 (30.5) 26 (15.9) Leading to discontinuation of any study drug 6 (11.1) 1 (1.9) 21 (12.8) 16 (9.8) Leading to death aa 1 (1.9) bb 1 (1.9) cc 3 (1.8) dd 4 (2.4) ee Immune-related TRAEs 18 (33.3) 10 (18.5) 54 (32.9) 29 (17.7) Grade ≥3 5 (9.3) 3 (5.6) 16 (9.8) 10 (6.1) VEGF-related TRAEs 16 (29.6) 7 (13.0) 57 (34.8) 26 (15.9) Grade ≥3 2 (3.7) 2 (3.7) 14 (8.5) 5 (3.0)
[Interpretation]
This safety analysis from the HARMONI study evaluates the tolerability of ivonescimab (a PD-1/VEGF bispecific antibody) combined with chemotherapy in patients with EGFR-mutated NSCLC, stratified by the presence of brain metastases.
Core Findings and Trends
1. Comparable Safety Profile Across Subgroups The most critical signal is that the safety profile of ivonescimab + chemotherapy remains consistent regardless of brain metastasis status. Treatment-related adverse events (TRAEs) occurred at similar frequencies in patients with brain metastases (88.9%) and those without (97.0%). This suggests that the presence of intracranial disease does not inherently predispose patients to a higher incidence of systemic toxicity when treated with this combination.
2. Incremental Toxicity of Ivonescimab The addition of ivonescimab to chemotherapy resulted in a predictable increase in high-grade and serious toxicities compared to the placebo + chemotherapy arms:
- Grade $\geq$ 3 TRAEs: Increased by approximately 6–8% in the ivonescimab arms (reaching ~48–51%).
- Serious TRAEs: There was a notable increase in serious events, particularly in patients without brain metastases (30.5% for ivonescimab vs. 15.9% for placebo).
- Discontinuation: Rates of drug discontinuation were higher in the ivonescimab arms (11–13%) compared to placebo, though fatal TRAEs remained low and balanced across all groups (~2%).
3. Mechanism-Specific Adverse Events Ivonescimab’s dual inhibition of PD-1 and VEGF is reflected in the specific toxicity signals:
- Immune-related TRAEs (irTRAEs): These were nearly doubled in the ivonescimab arm (~33% vs. ~18% in placebo). However, Grade $\geq$ 3 irTRAEs remained relatively infrequent (<10%).
- VEGF-related TRAEs: Consistent with VEGF inhibition, these events were more common in the ivonescimab groups (~30–35% vs. ~13–16%). Notably, Grade $\geq$ 3 VEGF-related events were higher in the "without brain metastases" subgroup (8.5%) compared to the "with brain metastases" subgroup (3.7%). This suggests that the risk of severe VEGF-mediated complications (like hemorrhage or hypertension) is not increased by the presence of brain lesions.
Professional Conclusion
The data indicate that ivonescimab + chemotherapy has a manageable safety profile. While the combination increases the frequency of serious and mechanism-driven (immune/VEGF) toxicities compared to chemotherapy alone, these events do not appear to be exacerbated by brain metastases. For clinicians, this supports the use of ivonescimab in the EGFR-TKI resistant population even when intracranial disease is present, as the "safety cost" of adding the bispecific antibody is consistent across these high-risk subgroups.
Chemo, chemotherapy; ivo, ivonescimab; TRAE, treatment-related adverse event; VEGF, vascular endothelial growth factor.
ddTRAEs leading to death were 1 patient with disease progression, 1 patient with multiple organ dysfunction, and 1 patient with gastrointestinal hemorrhage and pulmonary embolism. eeTRAEs leading to death were pneumonitis, myocardial infarction, cerebrovascular accident, and embolic stroke, each in 1 patient.
Table 3. Most Common TRAEs (≥20% in Either Treatment Group)
Patients with brain metastases Patients without brain metastases Ivo + chemo n=54 Placebo + chemo n=54 Ivo + chemo n=164 Placebo + chemo n=164 Ivo + chemo n=164 Placebo + chemo n=164 Ivo + chemo n=164 Characteristic, aa n (%) Any grade Grade ≥3 Any grade Grade ≥3 Any grade Grade ≥3 Any grade Anemia 22 (40.7) 5 (9.3) 28 (51.9) 7 (13.0) 85 (51.8) 17 (10.4) 95 (57.9) White blood cell count decreased 21 (38.9) 8 (14.8) 20 (37.0) 6 (11.1) 77 (47.0) 20 (12.2) 76 (46.3) Neutrophil count decreased 19 (35.2) 10 (18.5) 22 (40.7) 10 (18.5) 74 (45.1) 32 (19.5) 70 (42.7) AST increased 18 (33.3) 1 (1.9) 12 (22.2) 1 (1.9) 50 (30.5) 1 (0.6) 45 (27.4) Nausea 17 (31.5) 1 (1.9) 17 (31.5) 0 44 (26.8) 2 (1.2) 44 (26.8) Platelet count decreased 16 (29.6) 7 (13.0) 13 (24.1) 2 (3.7) 55 (33.5) 20 (12.2) 48 (29.3) ALT increased 14 (25.9) 2 (3.7) 12 (22.2) 1 (1.9) 45 (27.4) 3 (1.8) 50 (30.5) Decreased appetite 13 (24.1) 0 13 (24.1) 0 41 (25.0) 0 26 (15.9) Vomiting 11 (20.4) 1 (1.9) 6 (11.1) 0 34 (20.7) 2 (1.2) 35 (21.3)
[Interpretation]
This analysis interprets Table 3 from the HARMONI study, focusing on the safety profile of ivonescimab (a PD-1/VEGF bispecific antibody) plus chemotherapy in patients with EGFR-mutated NSCLC who have progressed on TKI therapy.
Core Findings and Critical Signals
1. Dominance of Hematologic Toxicities The most frequent treatment-related adverse events (TRAEs) across all cohorts are hematologic, specifically anemia, decreased white blood cell (WBC) count, and decreased neutrophil count.
- Anemia was the most common event, occurring in 40.7% to 57.9% of patients.
- Grade ≥3 Neutropenia (neutrophil count decreased) represented the most significant high-grade toxicity, peaking at 19.5% in the ivonescimab arm. These signals are largely consistent with the known safety profile of platinum-based chemotherapy backbones.
2. Safety Consistency Across Brain Metastases (BM) Subgroups A critical takeaway is that the presence of brain metastases at baseline did not exacerbate the systemic toxicity profile.
- For patients receiving ivonescimab + chemo, the incidence of "Any Grade" anemia was actually numerically lower in those with BM (40.7%) compared to those without BM (51.8%).
- High-grade (Grade ≥3) events remained relatively stable between the two subgroups, suggesting that ivonescimab can be administered to patients with intracranial disease without additional safety concerns regarding common TRAEs.
3. Ivonescimab vs. Placebo Comparison The addition of ivonescimab to chemotherapy did not result in a disproportionate increase in common toxicities compared to the placebo + chemotherapy control arms.
- In the BM+ group, the rates of nausea (31.5%) and decreased appetite (24.1%) were identical between the ivonescimab and placebo arms.
- Platelet count decrease showed a slight increase in the ivonescimab arm (29.6% vs 24.1% in BM+ patients), which may be a subtle signal of VEGF inhibition, but the overall delta remains narrow.
4. Low Incidence of High-Grade Hepatic and GI Distress While AST/ALT elevations and gastrointestinal issues (nausea/vomiting) were common (ranging from 20% to 33%), they were predominantly Grade 1 or 2. Grade ≥3 AST/ALT elevations were rare (≤3.7%), indicating that the combination is well-tolerated from a hepatic and gastrointestinal standpoint.
Professional Conclusion
The data in Table 3 indicates that the safety profile of ivonescimab plus chemotherapy is manageable and predictable. The toxicity is primarily driven by the chemotherapy component, with no evidence that ivonescimab significantly increases the burden of common TRAEs. Most importantly, the safety profile remains consistent regardless of brain metastasis status, supporting the potential use of this bispecific antibody in patients with intracranial disease—a population often considered higher risk in clinical settings. These findings, paired with the efficacy data mentioned in the conclusions, suggest a favorable benefit-risk ratio for this novel treatment modality.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; Chemo, chemotherapy; ivo, ivonescimab; TRAE, treatment-related adverse event. Events are listed according to decreasing incidence in the 'Any grade' column of the ivonescimab + chemotherapy treatment arm with brain metastases present.
CONCLUSIONS
• Ivonescimab + chemotherapy treatment was associated with numerically longer intracranial PFS benefit and a lower incidence of intracranial progression compared with placebo + chemotherapy in patients with and without brain metastases at baseline
- The safety profile of ivonescimab + chemotherapy was generally consistent with the known profiles of PD-L1 and VEGF pathway inhibition combined with chemotherapy, 7−97−9 and no new safety signals were identified across baseline brain metastases subgroups
• The intracranial benefit of adding ivonescimab to standard of care in patients with NSCLC regardless of brain metastases at baseline suggests a potential role for this novel treatment modality in the subset of patients with EGFR-mutated NSCLC progressed after third-line TKI
ACKNOWLEDGMENTS
The authors thank the participants and their caregivers, the investigators, and the site staff who have been and are participating in this study. This study is sponsored by Summit Therapeutics, Inc., and Akeso Biopharma funded data collection in China. Medical writing assistance was provided by MEDiSTRAVA (San Francisco, CA, USA) and was funded by Summit Therapeutics.
REFERENCES
1. Zhong T, et al. J Immunother Cancer. 2022;10(suppl 2):abstract 521 2. Fang W, et al. JAMA. 2024;332(7):561-570.
3. Zhang L, et al. Presented at the Society for Immunotherapy of Cancer (SITC) 40th Anniversary Annual Meeting; November 5-9, 2025; National Harbor, MD, USA. 4. PR Newswire. Published June 1, 2024. Accessed January 23, 2026. https://www.prnewswire.com/news-releases/ivonescimab-in-combination-with-chemotherapy-approved-in-china-by-nmpa-for-2l-egfrm-nsclc-based-on-harmoni-a-clinical-trial-positive-trend-observed-in-overall-survival-towards-ivonescimab-plus-chemotherapy-302161109.html. 5. ClinicalTrials.gov. NCT06396065. Accessed February 5, 2026. https://clinicaltrials.gov/study/NCT06396065?term=NCT06396065&rank=1. 6. Goldman JW, et al. Presented at the World Congress on Lung Cancer (WCLC); September 6-9, 2025; Barcelona, Spain. 7. Socinski MA, et al. N Engl J Med. 2018;378(24):2288-2301. 8. Park S, et al. J Clin Oncol. 2024;42(11):1241-1251. 9. Lu S, et al. Lancet Respir Med. 2023;11(7):624-636.
DISCLOSURES
Xiuning Le declares consulting/advisory fees from AbbVie, Abion, Akeso, Allist, ArriVent Biopharma, AstraZeneca, Avistone, Bayer, BioNTech, BlossomHill, Boehringer Ingelheim, Bristol Myers Squibb, Daiichi Sankyo, Dizal, Eli Lilly, EMD Serono (Merck KGaA), Hengrui, Innovent, Johnson & Johnson (Janssen), Merck, Novartis, Pfizer, Regeneron, Summit, SystImmune, Taiho, and Teligene; and research funding to institution from ArriVent Biopharma, Dizal, Eli Lilly, EMD Serono, Johnson & Johnson (Janssen), Regeneron, Takeda, Teligene, and Thermo Fisher.
Ivonescimab is an investigational therapy not approved by any regulatory authority other than China’s National Medical Products Administration (NMPA).
CONTACT INFORMATION
Please contact the presenting author, Xiuning Le, at xle1@mdanderson.org for questions or comments.
Copies of this poster obtained through QR (Quick Response) and/or text key codes are for personal use only and may not be reproduced without written permission of the authors. By use of the QR Code, you agree to the Summit's Privacy Notice, located at smmttx.com.
Presented at the 2026 European Lung Cancer Congress (ELCC); March 25-28, 2026; Copenhagen, Denmark