At the AACR 2026 Annual Meeting, Junshi Biosciences made a significant impact by presenting multiple heavy-hitting pipelines under its "Immuno-Oncology 2.0 (IO 2.0)" strategy. The latest clinical and preclinical data for two "star pipelines"—the PD-1/VEGF bispecific antibody JS207 and the EGFR/HER3 bispecific ADC JS212—were particularly eye-catching, demonstrating Junshi’s explosive potential in next-generation cancer therapies.
JS207 (PD-1/VEGF Bispecific Antibody)
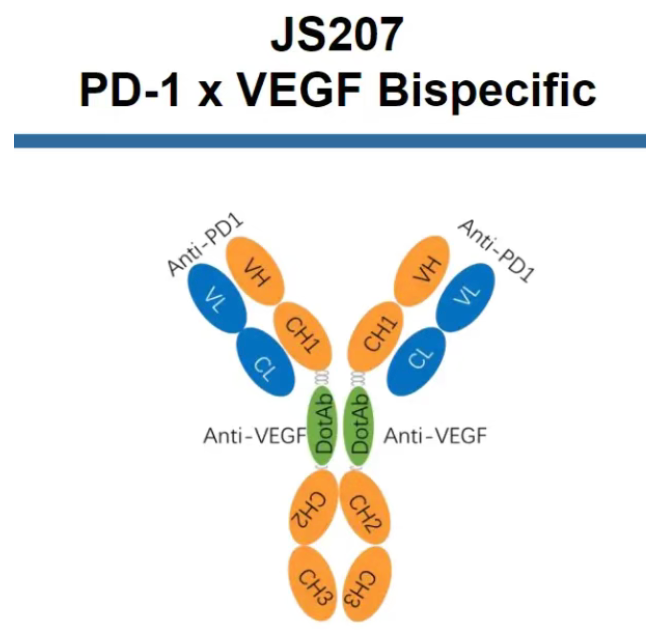
JS207 is a recombinant humanized anti-PD-1/VEGF bispecific antibody independently developed by Junshi Biosciences. At this year's AACR, preliminary results from two Phase 2 clinical trials evaluating JS207 as a first-line (1L) treatment were announced:
1. Combination with JS007 (anti-CTLA-4) for 1L Advanced Hepatocellular Carcinoma (HCC)
In this Phase 2 study involving patients with unresectable or metastatic HCC who had not received prior systemic therapy, the dual-immunotherapy combination showed a powerful synergistic effect.
-
Impressive Efficacy: As of March 20, 2026, among 22 evaluable patients, the Objective Response Rate (ORR) reached 45.5%, while the Disease Control Rate (DCR) was as high as 86.4%.
-
Excellent Safety: The treatment was well-tolerated, with no Dose-Limiting Toxicities (DLTs) observed during the safety lead-in period. This innovative combination is expected to bring new hope to patients with advanced liver cancer.
2. Combination with Chemotherapy (XELOX) for 1L Metastatic Colorectal Cancer (mCRC)
Most colorectal cancers are classified as "cold tumors," making them traditionally difficult to treat with immunotherapy monotherapies.
-
Breaking through Cold Tumors: As of January 13, 2026, among 31 evaluable patients, 22 achieved Partial Response (PR) and 8 achieved Stable Disease (SD). The ORR reached a staggering 71.0%, with a DCR of 96.8%.
-
Long-term Benefit: The longest Duration of Response (DOR) has reached 8 months and is still ongoing. This study provides critical clinical evidence for the potential of "Dual-target IO + Chemotherapy" in immune-cold tumors.
JS212 (EGFR/HER3 Bispecific ADC): First-in-Human Results
A heavyweight entry in Junshi’s ADC portfolio, JS212 is a recombinant humanized EGFR/HER3 bispecific ADC. Since EGFR and HER3 are highly expressed in various solid tumors (such as lung, breast, and head and neck cancers) and HER3 is often involved in resistance to targeted and chemotherapies, JS212 was designed to overcome drug resistance and broaden the benefit for patients.
1. Preclinical Data: Tackling Resistance Head-on
In several Cell-Derived Xenograft (CDX) models, JS212 demonstrated broad-spectrum anti-tumor activity superior to control drugs. Most notably, it showed potent efficacy in models resistant to Osimertinib, Patritumab deruxtecan, and BL-B01D1.
2. FIH Phase 1/2 Data: 100% DCR in High-Dose Groups
The CT128 trial, designed to evaluate the safety and efficacy of JS212 in patients with advanced solid tumors, delivered highly promising results:
-
Optimal Safety: As of March 26, 2026, 63 patients were enrolled. The treatment was extremely well-tolerated, and the Maximum Tolerated Dose (MTD) has not yet been reached.
-
Dose-Dependent Efficacy: Tumor responses were observed at doses as low as 1.8 mg/kg. In the high-dose groups (4.2 mg/kg and 4.6 mg/kg), the ORR reached 44.4% and 50.0% respectively, with both groups achieving a perfect 100.0% DCR.
-
Multi-Cancer Potential: An ORR of 50.0% was observed in HER2-negative breast cancer, and 38.9% in esophageal squamous cell carcinoma (ESCC). Even as a monotherapy, JS212 demonstrates immense clinical value.
Summary
Junshi Biosciences is currently accelerating clinical exploration for these two candidates across multiple indications and combination therapies.
Dr. Jianjun Zou, General Manager and CEO of Junshi Biosciences, commented:
"At this year's AACR, we not only reported data for two combination therapies featuring our bispecific JS207 but also released First-in-Human results for our bispecific ADC JS212. Both have demonstrated extremely exciting clinical profiles. With their superior efficacy, excellent activity in treatment-resistant populations, and broad-spectrum potential, these two products represent our next-generation flagship pipeline."