While GLP-1 receptor agonists have fundamentally disrupted the obesity treatment landscape, clinical bottlenecks remain: poor tolerance for lifelong injections, significant challenges in persistence, and the "weight rebound" effect post-discontinuation. These factors currently limit the long-term clinical utility of these therapies.
Recently, Eli Lilly published two landmark studies in The Lancet and Nature Medicine: Surmount-Maintainand Attain-Maintain. Together, these studies signal a strategic shift in the industry—moving from "acute weight loss" to "long-term maintenance" through dosage tapering and injectable-to-oral switching.
I. Surmount-Maintain: High-Dose Maintenance vs. Low-Dose Tapering
Published in The Lancet, the Surmount-Maintain study investigated the impact of dose reduction on efficacy after achieving significant weight loss with Zepbound (Tirzepatide).
-
Baseline: Participants achieved an average weight loss of approximately 50 lbs after a 60-week induction phase with the maximum tolerated dose (MTD) of Tirzepatide.
-
Study Arms:
- High-Dose Group: Continued the MTD for an additional 12-month maintenance period.
- Low-Dose Group (5mg): Tapered down to a 5mg dose for maintenance.
-
Key Findings:
- The MTD group achieved an additional weight loss of 2 lbs during the maintenance phase, demonstrating stable, incremental efficacy.
- The 5mg group experienced a noticeable rebound, with an average weight gain of 12 lbs.
-
R&D Insight: While the 5mg dose still outperformed placebo (which typically sees more drastic rebound), the data suggests that for GIP/GLP-1 dual agonists, maintaining high receptor occupancy is critical to preventing the re-accumulation of adipose tissue.
II. Attain-Maintain: Seamless Switch from "Injectable" to "Oral"
The Attain-Maintain study, published in Nature Medicine, carries even greater strategic weight. It validated the feasibility of using Lilly’s newly launched oral small-molecule GLP-1 RA, Foundayo (Orforglipron), as a maintenance therapy to replace injectables.
This study simulates a high-demand, real-world scenario: Can patients transition away from chronic injections once they reach their target weight?
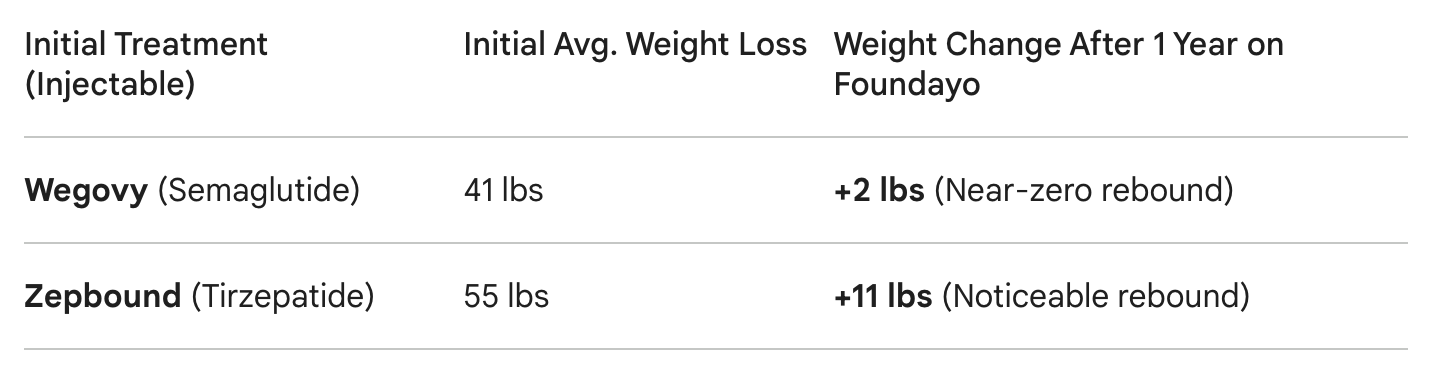
Analysis:
-
The Wegovy Switch: The transition was remarkably successful, with almost no rebound. This indicates that a potent small-molecule oral GLP-1 RA can effectively "pick up the baton" from an injectable single-target agonist like Semaglutide.
-
The Zepbound Switch: The 11 lb rebound is significant but must be contextualized by the larger initial weight loss (55 lbs vs. 41 lbs). This suggests a "potency gap" when switching from a dual-agonist (GIP/GLP-1) to a single-target (GLP-1) oral drug, highlighting a new R&D challenge for future multi-target oral therapies.
III. R&D Perspective: The Clinical & Commercial Logic of Maintenance
The publication of these studies marks the entry of obesity treatment into the era of "Sequential Therapy":
-
Adherence-Driven Formulations: While injectables offer high efficacy, "needle phobia" and cold-chain requirements limit penetration. As a non-peptide molecule, oral Orforglipron offers superior bioavailability and lower manufacturing costs, making it the ideal candidate for maintenance.
-
Balancing Economics and Safety: Long-term use of MTD injectables carries high costs and increased risks of GI-related adverse events. A "High-dose induction + Low-dose/Oral maintenance" model significantly improves the long-term safety window and reduces the economic burden on healthcare systems.
-
Preventing Metabolic "Set-Point" Reset: Obesity is a chronic disease. Post-discontinuation rebound is essentially the body’s metabolic self-correction. Lilly’s data proves that continuous pharmacological intervention—even at a tapered level—is necessary to stabilize the metabolic threshold.
Conclusion
In the second half of the "Weight Loss Race," the competitive edge is shifting from "how fast can you lose it" to "how well can you keep it off." Through the Surmount and Attain series, Lilly is constructing a closed-loop matrix ranging from the most potent injectables to convenient oral solids.
For R&D professionals, the next decade of metabolic research will center on optimizing switch timing and bridging the efficacy gap between multi-target induction and single-target maintenance.