On April 15, The New England Journal of Medicine (NEJM) published a landmark clinical study highlighting the remarkable efficacy of the novel oral HER2 inhibitor, Zongertinib, as a first-line treatment for patients with HER2-mutant non-small cell lung cancer (NSCLC).
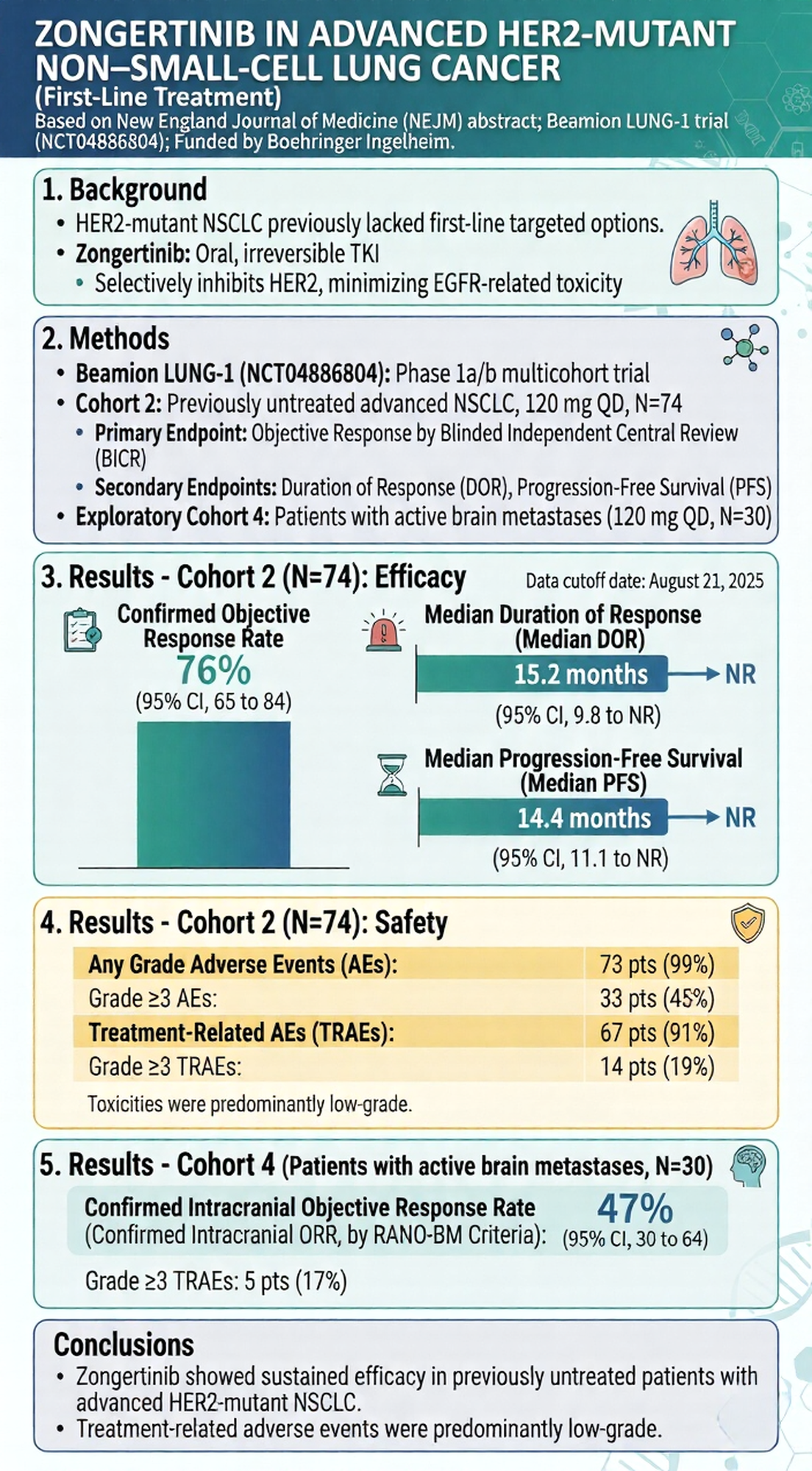
The study, based on the Beamion LUNG-1 trial, demonstrated that Zongertinib achieved an objective response rate (ORR) of up to 76% in treatment-native patients, along with good safety profile and strong intracranial anti-tumor activity.
Approximately 2%–4% of NSCLC patients harbor HER2 (ERBB2) mutations, most commonly exon 20 insertions in the tyrosine kinase domain (TKD). These mutations lead to constitutive activation of the HER2 protein, driving uncontrolled tumor growth and metastasis.
Zongertinib and Its Clinical Profile
Zongertinib is a novel, oral, irreversible, and highly selective HER2-targeted tyrosine kinase inhibitor (TKI) developed by Boehringer Ingelheim.
Renowned oncologist and investigator Dr. John V. Heymach once noted:
“Compared with previously tested agents, Zongertinib demonstrates approximately two orders of magnitude greater selectivity for HER2 mutations.”
This high level of selectivity enables the drug to precisely target and “lock onto” mutant HER2 while sparing wild-type EGFR, thereby maximizing anti-tumor efficacy and minimizing the adverse effects commonly associated with traditional TKIs.
Key Efficacy Results: Impressive First-Line Performance
The NEJM publication focuses on previously untreated patients with advanced HER2-mutant NSCLC.
- Objective Response Rate (ORR):
Zongertinib achieved an ORR of up to 76%, meaning nearly 80% of patients experienced significant tumor shrinkage. - Disease Control Rate (DCR):
An impressive 96% of patients achieved disease control, indicating that nearly all enrolled patients experienced stabilization or reduction of disease. - Rapid Onset of Action:
The median time to response was just 1.4 months, demonstrating a rapid therapeutic effect soon after treatment initiation. - Robust Intracranial Activity:
Brain metastases are a major cause of mortality in lung cancer. Zongertinib showed strong blood–brain barrier penetration, with intracranial disease control rates exceeding 80% in patients with brain metastases across study cohorts, offering new hope for this high-risk population.
Safety Advantages: Minimal Toxicity, No ILD Observed
Due to its high selectivity and minimal off-target EGFR inhibition, the incidence of severe diarrhea and rash was very low.
Importantly, no cases of treatment-related interstitial lung disease (ILD) or pneumonitis were observed in the current clinical cohorts. Compared to some antibody-drug conjugates (ADCs), Zongertinib not only eliminates the need for frequent hospital-based intravenous infusions but also alleviates concerns about potentially fatal pulmonary toxicity.
This favorable safety profile significantly improves patient quality of life, with only approximately 3% of patients discontinuing treatment due to adverse events.
A Milestone in Precision Oncology
Zongertinib is rapidly reshaping the treatment landscape for HER2-mutant lung cancer. Based on earlier data showing over 70% response rates and progression-free survival (PFS) exceeding 12 months in previously treated patients, the drug has already received accelerated approval from the U.S. FDA and conditional approval from China’s NMPA.
The newly published first-line data in NEJM has further led the FDA to grant Zongertinib Breakthrough Therapy Designation for first-line treatment of advanced HER2-mutant non-squamous NSCLC.
Experts widely believe that, given its compelling efficacy and favorable safety profile, Zongertinib has the potential to replace traditional chemotherapy and become the new gold-standard first-line therapy for patients with HER2-mutant lung cancer.