With the approval of Eli Lilly’s oral GLP-1 drug Orforglipron, the global obesity drug battlefield has expanded from injectables into the oral domain. Clinical development and commercialization of GLP-1 therapies have now entered a phase of full-scale, intense competition. Currently, the two most prominent drugs on the market are:
- Novo Nordisk’s oral version of Wegovy (semaglutide), approved by the U.S. Food and Drug Administration (FDA) in December 2025 for obesity treatment.
- Eli Lilly’s Orforglipron, which quickly followed and received rapid approval under the FDA’s innovative “Commissioner’s National Priority Voucher (CNPV)” pilot program, achieving approval in just 50 days in early April 2026 under the brand name Foundayo.
This article is based on the core Phase 3 clinical trials of these two oral weight-loss drugs—OASIS-1, OASIS-4, and ATTAIN-1—and provides a comparative analysis across multiple dimensions, including efficacy ceiling, safety and tolerability, pharmacokinetics, real-world adherence, and future commercial potential.
Overview of Core Clinical Trials
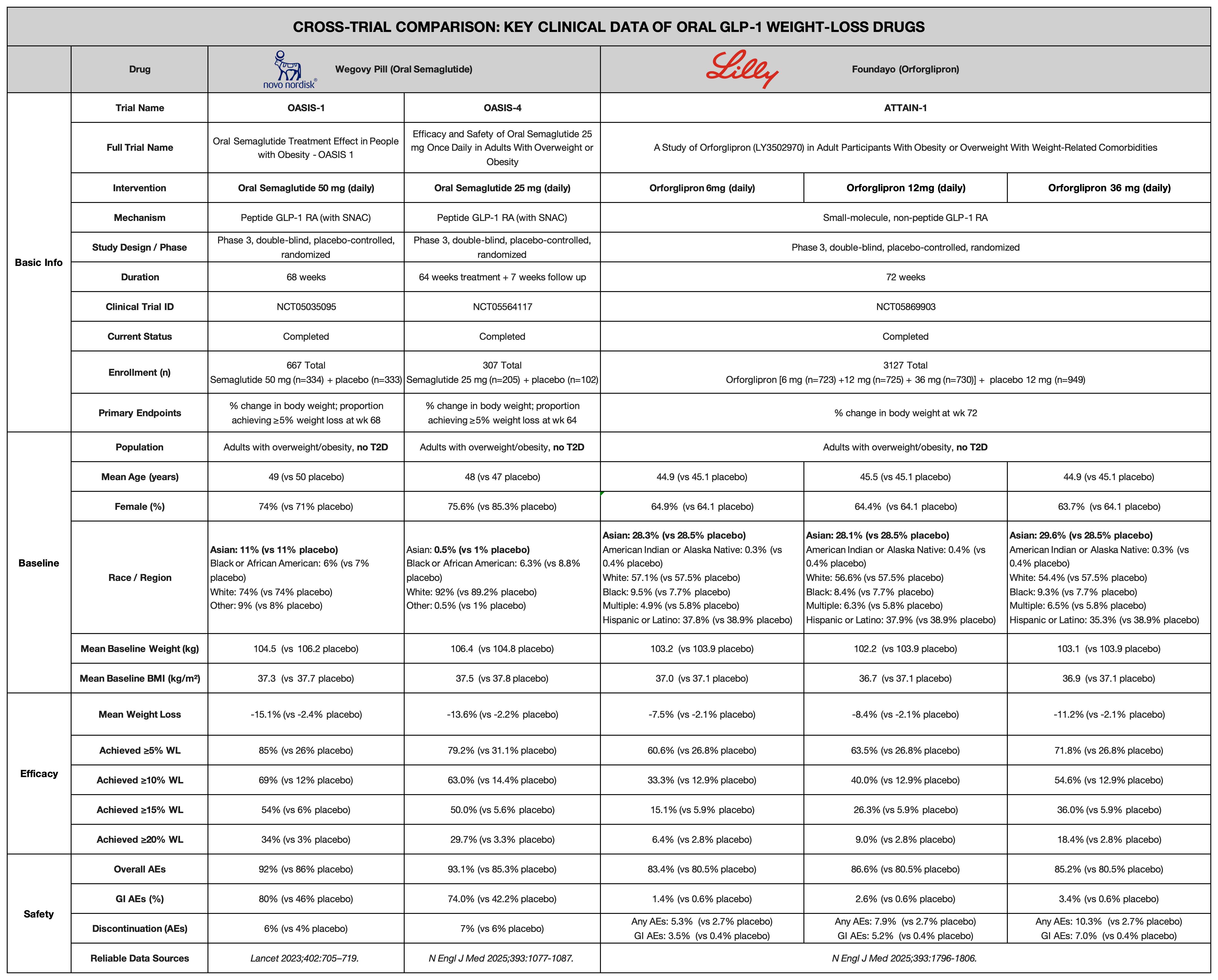
Considerations in Trial Selection
All three pivotal Phase 3 trials adopted multicenter, randomized, double-blind, placebo-controlled designs. They enrolled overweight or obese individuals without diabetes. All participants received standardized lifestyle interventions alongside drug treatment (typically including a 500 kcal/day caloric deficit and at least 150 minutes of physical activity per week).
Inclusion criteria were highly consistent across trials:
- Adults ≥18 years
- BMI ≥30 kg/m² (obese), or ≥27 kg/m² (overweight) with at least one weight-related comorbidity (e.g., hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease)
- Exclusion of patients with diagnosed type 1 or type 2 diabetes
- Documented history of unsuccessful dietary weight loss
OASIS-1 Trial
Designed to evaluate ultra-high-dose oral semaglutide (50 mg).
- Duration: 68 weeks
- Enrollment: 667 patients
- Randomization: 1:1 (semaglutide 50 mg vs. placebo)
- Dose escalation: Starting at 3 mg, increasing every 4 weeks (3 → 7 → 14 → 25 mg), reaching 50 mg maintenance at week 16
OASIS-4 Trial
Evaluated a more balanced and tolerable dose of oral semaglutide (25 mg).
- Duration: 64-week treatment + 7-week follow-up (71 weeks total)
- Enrollment: 307 patients
- Randomization: 2:1
- Dose escalation: 3 → 7 → 14 mg, reaching 25 mg maintenance at week 12
ATTAIN-1 Trial
A large-scale trial evaluating multiple dose levels of the small molecule Orforglipron.
- Duration: 72 weeks
- Enrollment: 3,127 patients
- Randomization: 3:3:3:4 (6 mg, 12 mg, 36 mg, placebo)
- The 36 mg group used a prolonged titration schedule starting at 1 mg, reaching 36 mg only by week 20
This extended titration reflects a deliberate strategy to mitigate anticipated gastrointestinal adverse effects.
Baseline Insights
Age and Gender Distribution
- ATTAIN-1 population (~45 years) was slightly younger than OASIS (~48–50 years).
- OASIS trials had a high proportion of women (73%–85%), typical for obesity trials.
- ATTAIN-1 capped female enrollment at 70%, resulting in ~64% female participation.
Since men generally experience less weight loss with incretin-based therapies, this gender difference may slightly disadvantage ATTAIN-1 in cross-trial comparisons.
Racial Diversity and Global Representation
- OASIS-4 was predominantly White (~90%), with <1% Asian participants.
- ATTAIN-1 showed strong global representation: ~29% Asian and ~55% White.
Such differences may impact drug metabolism, clearance, and fat distribution due to genetic and physiological variability.
Efficacy Comparison
OASIS-1 (50 mg Oral Semaglutide)
- Mean weight reduction at week 68: –15.1%
- Placebo-adjusted difference: –12.7%
This sets a new benchmark for oral weight-loss drugs and approaches the efficacy of injectable Wegovy (~14.9% in STEP-1).
OASIS-4 (25 mg Oral Semaglutide)
- Mean weight reduction: –13.6%
- Placebo-adjusted difference: –11.4%
Reducing the dose by half resulted in only ~1.5% loss in efficacy, suggesting a near-saturation point in receptor activation around 25 mg.
ATTAIN-1 (Orforglipron)
- 36 mg: –11.2% (placebo-adjusted –9.1%)
- 12 mg: –8.4%
- 6 mg: –7.5%
Conclusion on Efficacy
A clear gradient emerges:
- 11.2% (Orforglipron 36 mg)
- 13.6% (Oral semaglutide 25 mg)
- 15.1% (Oral semaglutide 50 mg)
This suggests that first-generation small-molecule agonists may still lag behind structurally optimized peptide drugs in receptor binding efficiency and central appetite regulation.
Additional Efficacy Considerations
Blood Pressure and Fat Distribution
Both drugs significantly reduced systolic blood pressure and waist circumference, indicating visceral fat reduction.
Lipid Metabolism
Orforglipron significantly reduced triglycerides and non-HDL cholesterol, indicating reduced atherosclerotic risk.
Physical Function and Quality of Life
In OASIS-4, oral semaglutide significantly improved physical function scores (IWQOL-Lite-CT).
- 55.3% achieved clinically meaningful improvement vs. 34.8% in placebo
Safety and Tolerability
Oral Semaglutide
- OASIS-1: 92% adverse events (AEs), 80% GI-related
- OASIS-4: GI AEs reduced to 74%
Most were mild-to-moderate and transient, with good long-term tolerability.
Orforglipron (ATTAIN-1)
- Overall AE rate: 83.4%–86.6%
- Discontinuation due to AEs: 5.3%–10.3% (vs. 2.7% placebo)
Despite careful titration, discontinuation rates remain a key weakness.
Safety Signals
- 5 mild pancreatitis cases reported (all in treatment group)
- Heart rate increased by 4.3–5.3 bpm
These require further long-term cardiovascular evaluation.
Final Summary
Based on ATTAIN-1 and the OASIS trials:
Oral semaglutide and Orforglipron are not merely competing in weight loss—they both demonstrate systemic metabolic benefits and improve overall patient outcomes.
They represent complementary strategies:
- Oral semaglutide (peptide-based, SNAC-enabled):
Sets the benchmark in efficacy and tolerability balance. Ideal for patients seeking maximal weight loss and capable of strict dosing adherence. - Orforglipron (small molecule):
Represents the future paradigm. While sacrificing some efficacy and tolerability, it offers unmatched convenience (no fasting required) and, critically, dramatically lower manufacturing costs and scalable supply.
This cost advantage positions Orforglipron to transform GLP-1 therapy from a premium treatment into a widely accessible global solution, particularly in low- and middle-income regions.
Outlook
Over the next decade, as the oral GLP-1 market rapidly expands, these two distinct molecular approaches—peptide and small molecule—will act as strategic complements, jointly driving metabolic disease treatment into an era of unprecedented scale and accessibility.