
Pancreatic cancer has long been dubbed the "King of Cancers" due to its devastating mortality rate, difficulty in early detection, and high recurrence. According to the American Cancer Society’s 2026 Cancer Statistics Report, the five-year survival rate remains a sobering 13%.
However, Memorial Sloan Kettering Cancer Center (MSK) recently released exhilarating follow-up data from a Phase 1 clinical trial of a customized mRNA vaccine. For patients who mounted an immune response to the vaccine, the survival rate 4 to 6 years post-surgery reached a staggering 87.5%, marking a major breakthrough in tumor immunotherapy.
About Autogene Cevumeran and Clinical Results
The Phase 1 trial was led by Dr. Vinod Balachandran, a physician-scientist at MSK and director of the Olayan Cancer Center. The team tested an experimental, personalized mRNA cancer vaccine called Autogene cevumeran (BNT122, RO7198457), co-developed by BioNTech and Genentech.
At the 2026 AACR Annual Meeting, Dr. Balachandran presented the latest long-term follow-up results. The trial enrolled 16 pancreatic cancer patients who, following tumor resection surgery, received the mRNA vaccine alongside standard chemotherapy and immune checkpoint inhibitors.
The clinical data showed a clear polarization, which precisely validates the vaccine's efficacy:
-
Immune Responders: Out of the 16 patients, 8 successfully had their immune systems "awakened" by the vaccine. Among these 8, 7 (87.5%) are still alive and well 4 to 6 years after surgery.
-
Non-Responders: Among the other 8 patients who did not produce a sufficient immune response, only 2 (25%) survived, with a median survival of 3.4 years.
"These early results are encouraging," said Dr. Balachandran. "They suggest that personalized mRNA vaccines can meaningfully stimulate the immune system in some pancreatic cancer patients, leading to sustained survival years after vaccination."
The Mechanism of the mRNA Vaccine
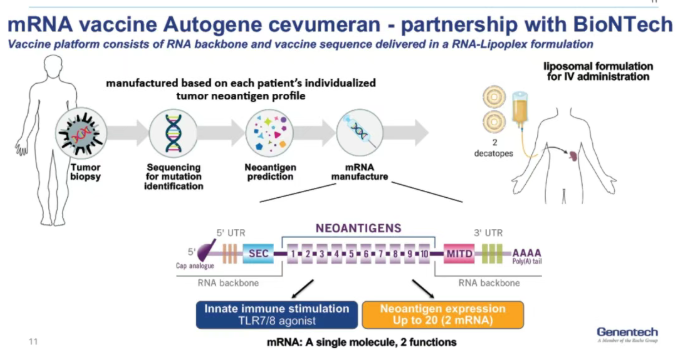
The secret to this vaccine's long-term efficacy lies in its complete customization and precise identification of "Neoantigens."
Historically, pancreatic cancer cells have been masters of disguise, preventing the immune system from recognizing them as "foreign." However, the research team discovered that some long-term survivors of pancreatic cancer possess special T cells capable of identifying neoantigens—mutations unique to the cancer—effectively stripping away the tumor's camouflage.
The production process for this trial is a feat of personalized medicine:
-
Genetic Sequencing: Tumor tissue removed during surgery is sent to BioNTech’s labs in Germany for deep genetic sequencing.
-
Precision Targeting: Scientists identify up to 20 tumor mutations (neoantigens) most likely to be flagged as foreign by the immune system.
-
Vaccine Synthesis: mRNA technology converts the genetic codes of these mutations into a vaccine. Once injected, the mRNA instructs the patient’s cells to produce these specific protein fragments.
-
Awakening the Army: Recognizing these proteins, the immune system generates a massive fleet of targeted T cells to hunt down and destroy any remaining pancreatic cancer cells.
Computational biologist Dr. Benjamin Greenbaum’s team found that responders developed not only CD8+ T cells (which kill cancer cells directly) but also CD4+ T cells (which maintain and boost the immune response). Remarkably, these anti-cancer T cells have been patrolling the patients' bodies for up to six years without showing signs of exhaustion.
Moving Beyond a "Death Sentence"

Donna Gustafson, the first patient to enroll in the trial in late 2019, was diagnosed with pancreatic cancer at 66. Despite the initial despair, she underwent surgery and received the mRNA vaccine tailored to her tumor's mutations. Six years later, she is not only alive but recently celebrated her 50th wedding anniversary on Mount Etna in Sicily. "To me, it's an absolute miracle," she remarked.
Looking Ahead
Building on the success of the Phase 1 trial, Genentech and BioNTech have launched a larger global Phase 2 clinical trial to validate the efficacy of autogene cevumeran across a broader patient population.