Recent presentations at major international medical conferences, including the European Association of Urology (EAU), highlighted promising Phase 1 clinical trial results for B440 in patients with metastatic urothelial cancer. B440 is an innovative oral cancer vaccine utilizing a Bifidobacterium vector, offering a novel therapeutic avenue for advanced malignancies.
Innovative Mechanism of Action
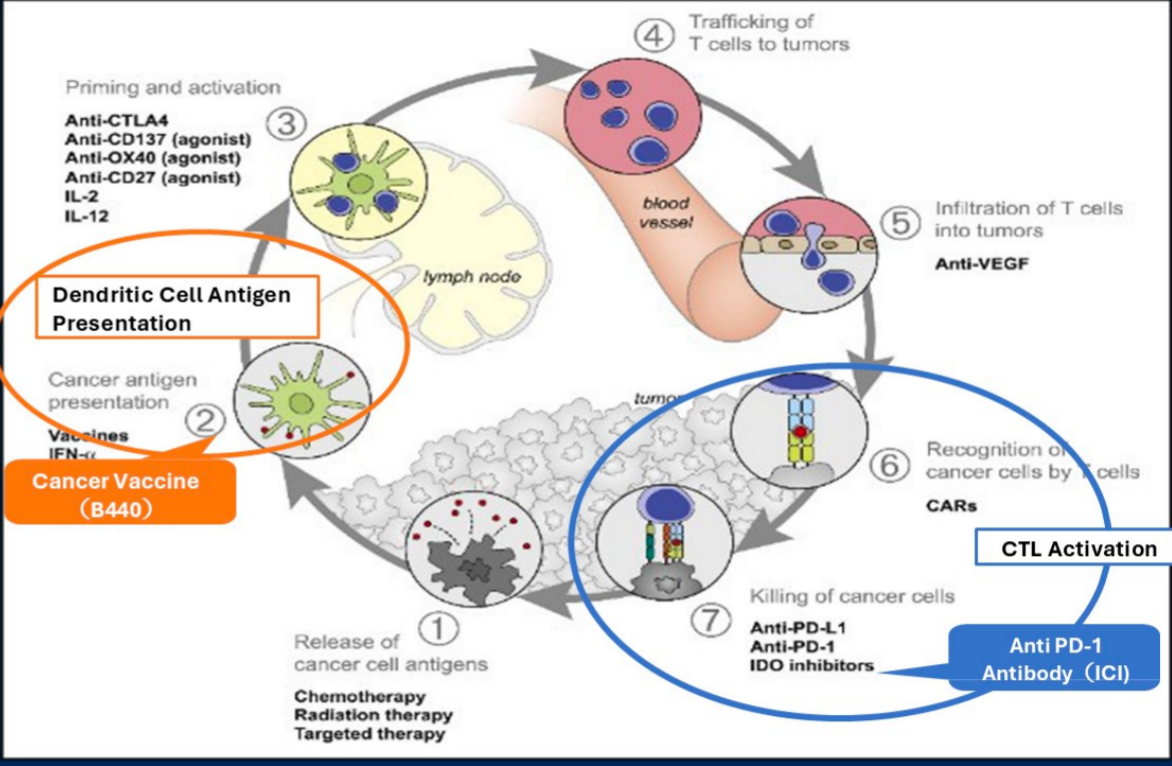
B440 is formulated using a recombinant gut bacterium, Bifidobacterium longum, engineered to express the human WT1 (Wilms' tumor 1) tumor-associated antigen. Upon oral administration, B440 efficiently delivers the WT1 antigen protein to dendritic cells within the gut-associated lymphoid tissue, specifically targeting Peyer's patches. This mucosal delivery process effectively induces a robust, tumor-specific cellular immune response driven by WT1-specific T cells.
Clinical Study Design and Findings This Phase 1 open-label, single-arm study evaluated the safety, immunogenicity, and preliminary efficacy of B440 in heavily pretreated patients with metastatic urothelial cancer whose disease had progressed after all standard therapies, including cytotoxic chemotherapy, PD-1/PD-L1 inhibitors, and antibody-drug conjugates.
Key findings from the study include:
-
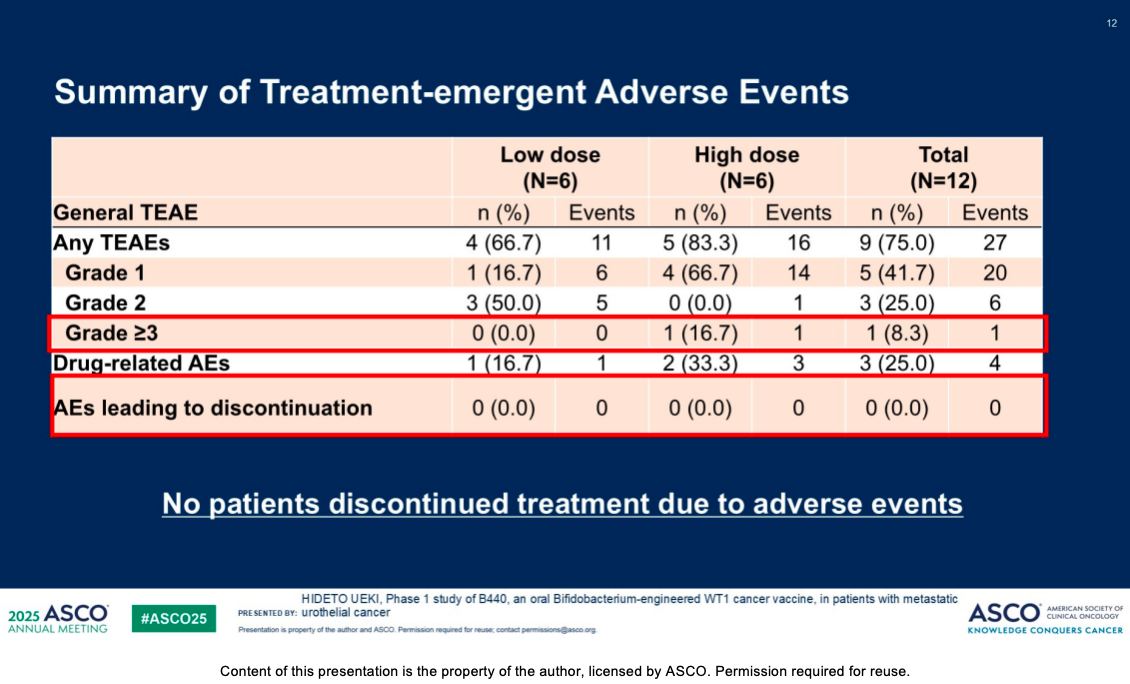
Favorable Safety Profile: B440 was exceptionally well-tolerated, with no dose-limiting toxicities (DLTs) observed at administered doses up to 1,600 mg.
-
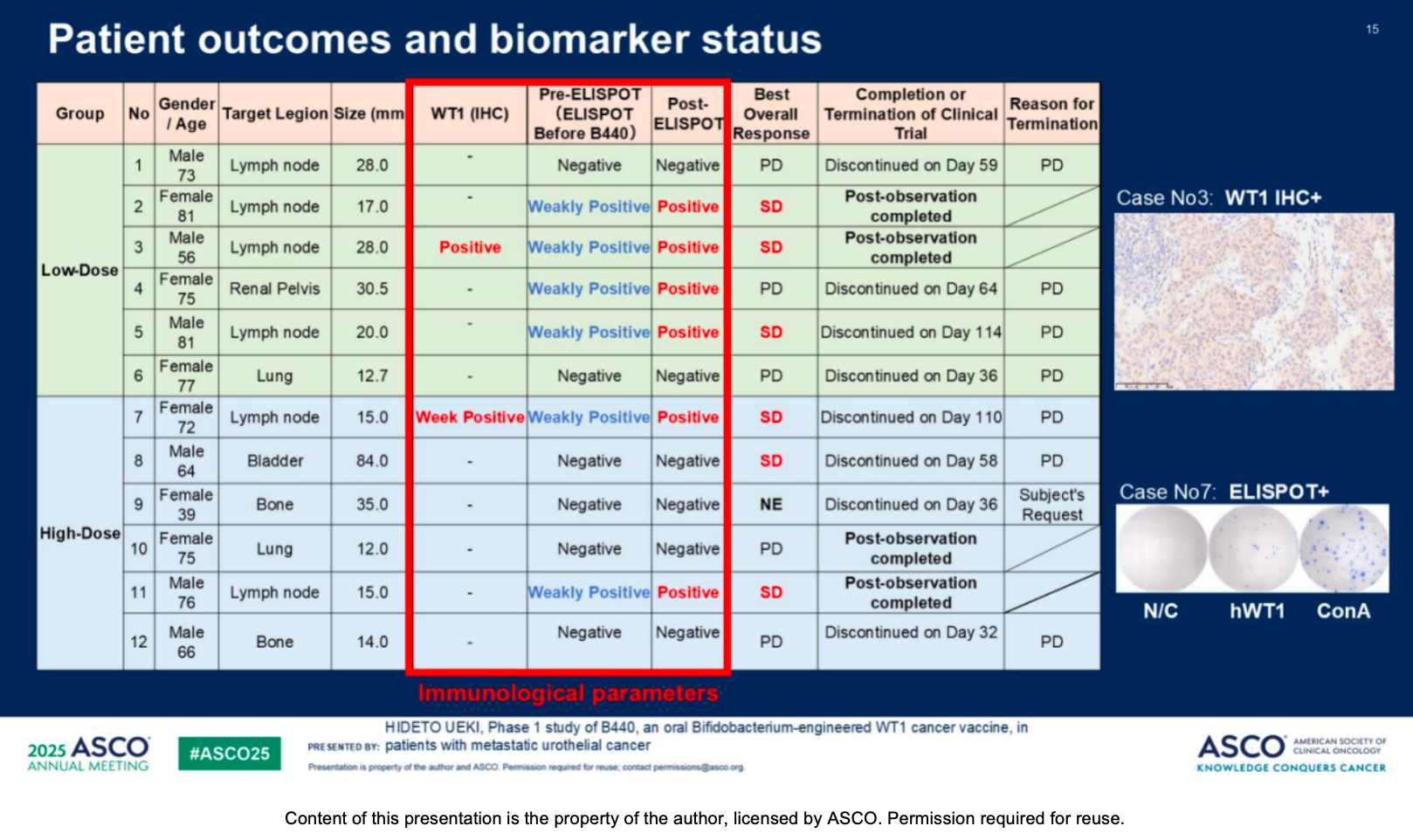
Promising Efficacy: The disease control rate (DCR) stood at 50%, with six patients achieving stable disease (SD) as their best overall response.
-
Strong Immunogenicity: ELISPOT analysis confirmed robust WT1-specific T-cell induction in six patients. Among these ELISPOT-positive individuals, early tumor shrinkage was noted, with maximum lesion reductions of -100%, -49%, and -32.7% from baseline.
-
Synergistic Potential: The induction of WT1-specific cellular immunity directly correlated with improved progression-free survival (PFS) during B440 therapy. Furthermore, it enhanced subsequent anti-tumor responses upon patient rechallenge with pembrolizumab (an anti-PD-1 antibody).
Future Perspectives
The highly encouraging data from this Phase 1 study suggest that B440 can successfully enhance the immunogenicity of urothelial tumors, potentially overcoming resistance and leading to durable disease control. These findings strongly support the further investigation of B440 in larger, randomized clinical trials and its strategic combination with other immunotherapies for WT1-expressing cancers.