April 23, 2026 — The FDA has granted accelerated approval to Regeneron’s innovative gene therapy, Otarmeni (lunsotogene parvec-cwha). This marks the world’s first AAV-vector gene therapy for hereditary deafness, moving from BLA submission to approval in a staggering 61 days.
In a landmark move, Regeneron announced that it will provide this breakthrough medication free of charge in the United States.
The OTOF Gene and the "Silent World"
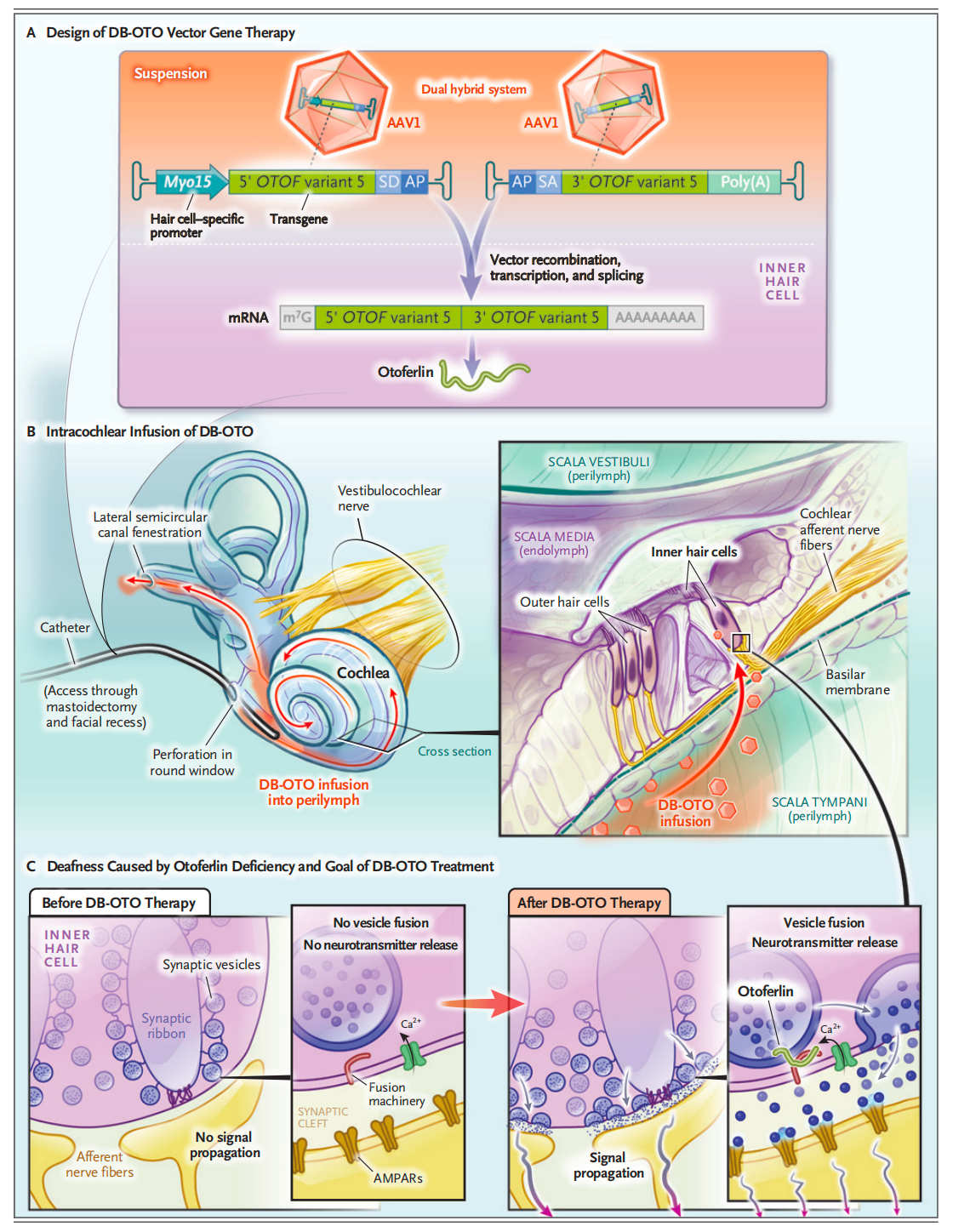
Hearing loss is one of the most common sensory deficits globally; in cases of congenital deafness, approximately half are caused by genetic mutations. Otarmeni is specifically indicated for children and adults with severe-to-profound sensorineural hearing loss (>90 dB HL at any frequency) caused by biallelic pathogenic variants in the OTOF (Otoferlin) gene.
-
Function: The OTOF gene encodes Otoferlin, a protein essential for inner ear hair cells to convert sound signals into neural impulses and transmit them to the auditory nerve.
-
The Defect: When this gene is mutated, the physical structure of the ear remains intact, but the brain receives no sound signals from the inner ear.
This is an extremely rare condition, affecting approximately 20 to 50 newborns in the U.S. each year. Previously, no disease-modifying drugs existed for OTOF-related deafness. The only option was cochlear implantation, which cannot fully replicate natural hearing. Due to auditory deprivation, many children miss the "golden window" for language development, facing lifelong speech and cognitive delays.
Clinical Data: One Injection to Restore Natural Hearing
Otarmeni is a dual adeno-associated virus (AAV1) vector gene therapy. Through a one-time, minimally invasive surgical procedure (an inner ear injection performed under general anesthesia, similar to a cochlear implant approach), it delivers functional OTOF genes directly to the hair cells, restoring otoferlin production and auditory signal transmission in situ.
Clinical data recently published in the New England Journal of Medicine (NEJM) from the CHORD Phase 1/2 single-arm trial (evaluating 24 patients aged 10 months to 16 years) showed:
-
Significant Improvement: Among 20 evaluable patients, 80% (16 patients) achieved a pure-tone average (PTA) threshold of ≤70 dB HL within 24 weeks—the clinical benchmark where reliance on cochlear implants is no longer mandatory.
-
Full Recovery: With longer follow-up, 42% of patients saw their hearing return to the normal range, with some able to hear whispers clearly.
-
Developmental Milestones: Beyond decibel thresholds, children achieved breakthroughs in speech perception. Without visual cues, some could identify bisyllabic words (e.g., "Mama," "Cookie") and react to distant speech in noisy environments, achieving true 24/7 natural hearing.
"Light Speed" Approval
Otarmeni is the sixth product—and the first gene therapy—approved under the FDA’s "Commissioner’s National Priority Voucher (CNPV)" pilot program. Due to its breakthrough efficacy and potential to address an unmet medical need, it received accelerated approval just 61 days after BLA submission.
"Today’s approval is a major milestone," said FDA Commissioner Dr. Marty Makary. "Through the CNPV pilot, the FDA has proven its ability to successfully and efficiently review complex applications like dual-vector gene therapies in record time, accelerating life-saving treatments for rare disease patients."
Free Access Across the U.S.
Gene therapies are notorious for multi-million dollar price tags. However, as part of an agreement with the U.S. government to lower drug costs, Regeneron announced it will provide Otarmeni entirely free of charge in the United States.
Dr. George Yancopoulos, Co-Founder, President, and Chief Scientific Officer of Regeneron, stated:
"Otarmeni is a giant leap for science and a testament to what happens when brilliant minds have the resources and freedom to tackle hard problems. When we saw the breathtaking data from the CHORD trial, the conversation turned to how we could ensure this science reaches as many people as possible. Our decision to provide Otarmeni for free in the U.S. underscores our conviction that the biopharmaceutical industry can, and should, be a true force for good in the world."