
The weight-loss drug market is becoming increasingly crowded, with GLP-1 currently the most sought-after target. From Novo Nordisk’s Semaglutide and Eli Lilly’s Tirzepatide to the newly launched oral GLP-1 Orforglipron, the fierce competition has expanded from subcutaneous injections to the oral market. These "miracle weight-loss drugs" often boast weight reduction effects of over 20%, leading countless individuals struggling with obesity to vote with their wallets. However, amidst this intense "weight-loss arms race," Roche is doubling down on Petrelintide, an Amylin-target drug with slightly lower efficacy.
Efficacy vs. Safety: The Great Debate
First, let's look at the comparison. An overview of the weight-loss efficacy and safety of Semaglutide vs. Tirzepatide vs. Petrelintide is shown in the chart below.
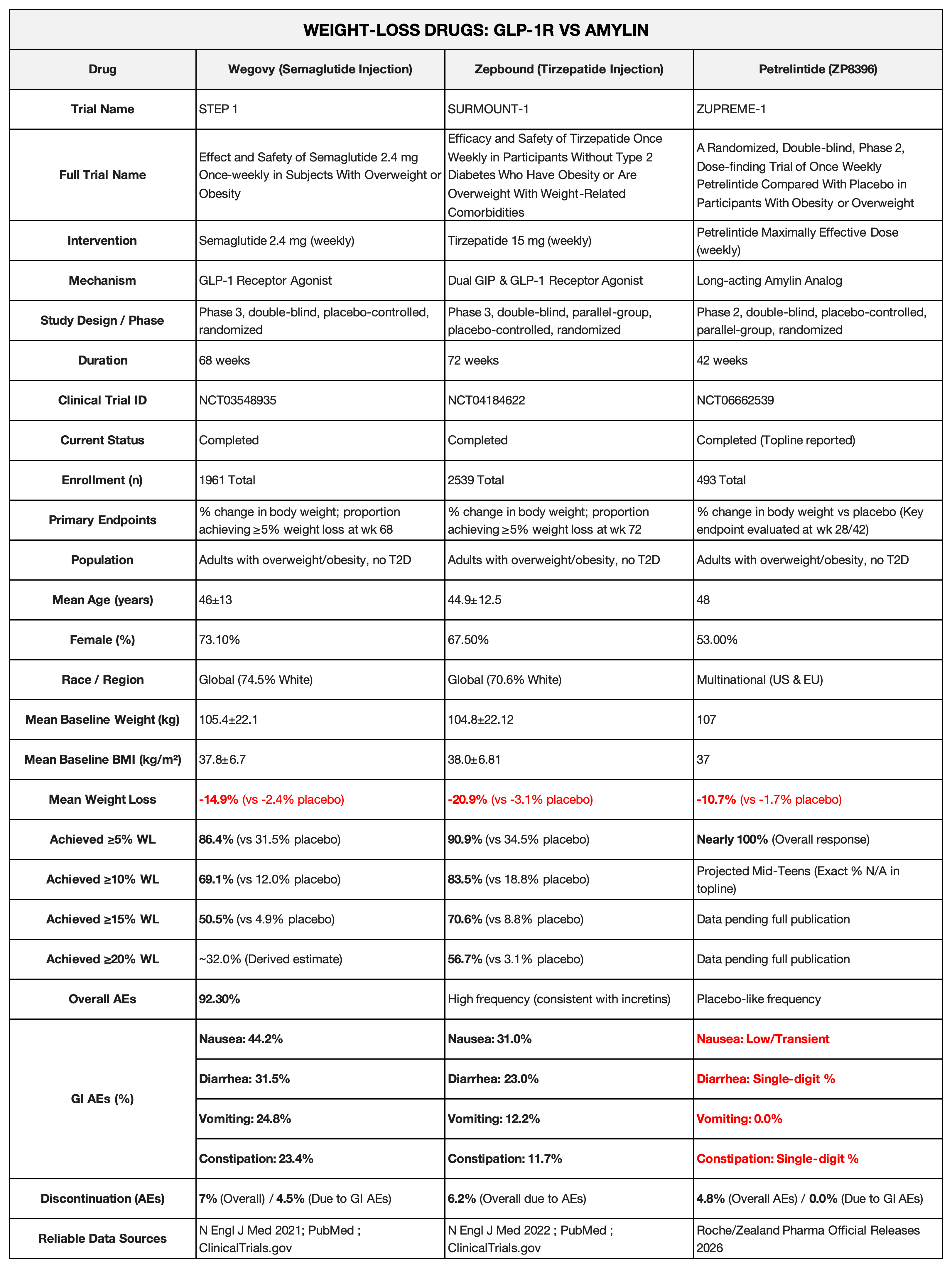
In March, Roche and Zealand Pharma jointly announced positive data from the Phase II clinical trial (ZUPREME-1) for Petrelintide, their co-developed novel long-acting Amylin analog.
According to Roche’s official press release on March 5, 2026, ZUPREME-1 was a clinical trial involving 493 overweight and obese patients (average BMI of 37 kg/m²). Data showed that after 42 weeks of treatment, subjects using Petrelintide achieved a maximum average weight loss of 10.7%, compared to just 1.7% in the placebo group.
To be fair, a 10.7% weight reduction is a solid performance in the obesity field. However, the capital markets were not impressed. Following the data release, Zealand Pharma’s stock price plummeted by as much as 32% in early trading.
The reason is simple: the competition is too cutthroat, and investor expectations are sky-high. Wall Street analysts had expected the drug to achieve at least a 12% weight loss. Compared to the current environment where GLP-1 drugs regularly hit 20% or more, sub-11% figures seem lackluster—becoming the primary driver for the market sell-off.
The "Ace in the Hole": Tolerability
Facing the stock price drop and external skepticism, Roche CEO Thomas Schinecker personally defended the drug during a recent Q1 earnings call. He admitted, "Indeed, the weight loss might not be as high as some other Amylin drugs. But weight loss is one thing; tolerability is another."
This statement hits the nail on the head. While GLP-1 drugs offer staggering results, their side effects leave many patients "suffering." Schinecker pointed out that for GLP-1/GIP drugs currently on the market, one-third of patients quickly discontinue treatment because they cannot tolerate severe gastrointestinal reactions like nausea and vomiting.
This is precisely Petrelintide’s "trump card"—extreme safety comparable to a placebo.
Consider these stunning safety figures released by Roche:
-
"Zero" Vomiting: In the treatment group reaching the highest effective dose, the number of vomiting cases was zero. No patients discontinued treatment due to gastrointestinal adverse events.
-
Ultra-low Discontinuation Rate: In the high-dose group, the discontinuation rate due to adverse events (AEs) was only 4.8%—lower than that of the placebo group (4.9%).
-
Minimal Nausea: Thanks to a gentle dosing regimen that escalates only every four weeks, 98% of subjects successfully reached the maintenance dose. More importantly, almost no patients reported nausea once the maintenance dose was achieved.
"If you want to chase the extreme 20%+ weight loss," Schinecker stated bluntly, "then you should choose GLP-1/GIP. But for patients who don’t want side effects and need a safer, highly tolerable option, Petrelintide is the better choice."
The Future of the Amylin Target
Is this "gentle" drug intended only for patients who are "afraid to vomit"? Roche has a much deeper strategic layout in mind.
-
Maintenance Therapy: Petrelintide is the perfect candidate for "maintenance phase" treatment. After patients reach their target weight using potent GLP-1 drugs, they often require long-term medication to prevent weight regain. With its extremely low side-effect profile, Petrelintide is an ideal relay drug for long-term weight management.
-
The Ultimate Combination Partner: Roche’s Chief Medical Officer, Dr. Levi Garraway, noted that Petrelintide possesses high chemical and physical stability (no fibrillation at neutral pH), allowing it to be easily combined with other peptide drugs. Roche is set to launch a Phase II trial later in 2026 combining Petrelintide with its potent GLP-1/GIP dual-agonist, CT-388. This "Potency + Tolerability" combo could potentially reshape the entire landscape of obesity treatment.
Additionally, results from ZUPREME-2, another Phase II trial targeting overweight/obese patients with Type 2 diabetes, are expected in the second half of 2026. This remains a space worth watching closely.