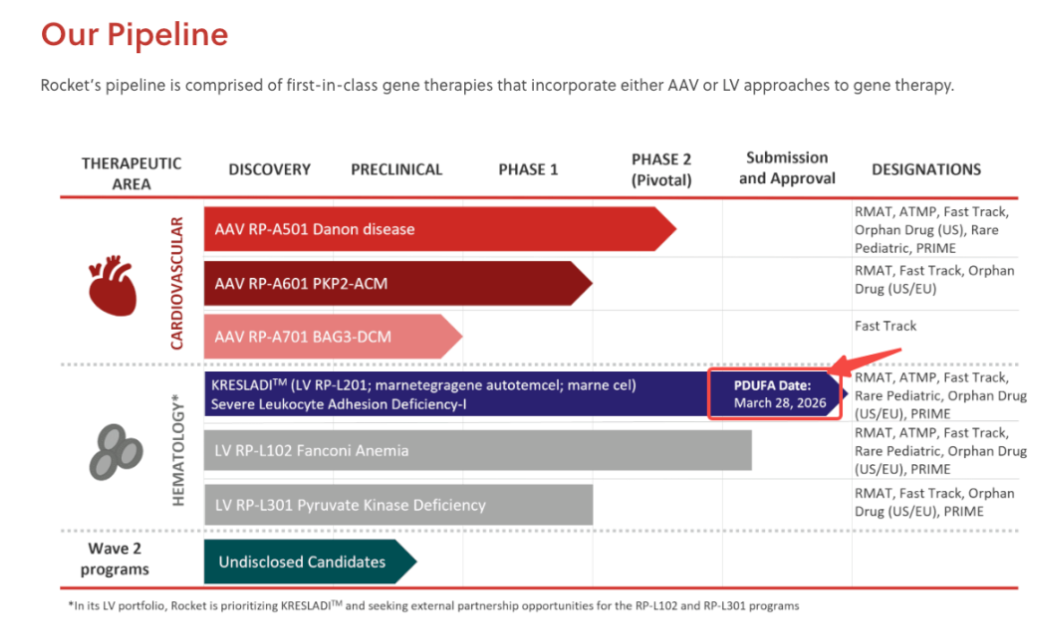
March 26 marks the final 48-hour sprint to the PDUFA date of March 28, 2026, when the U.S. FDA will issue its final decision on Rocket Pharma’s flagship gene therapy KRESLADI™ (marnetegragene autotemcel).
About KRESLADI
Leukocyte Adhesion Deficiency Type I (LAD-I) is an extremely rare, devastating, and fatal inherited immunodeficiency disorder. Caused by mutations in the ITGB2 gene, patients’ leukocytes fail to express the CD18 protein, preventing leukocytes from properly adhering to and penetrating blood vessel walls to reach sites of tissue infection. As a result, affected children have virtually no defense against even minor bacterial or fungal infections. Typical clinical manifestations include delayed umbilical cord separation after birth and recurrent life-threatening infections of the skin and mucous membranes.
Without allogeneic hematopoietic stem cell transplantation—previously the only curative option, which is limited by donor scarcity and severe transplant rejection risks—children with severe LAD-I face an almost 100% mortality rate in childhood.
Against this cruel "orphan disease", KRESLADI offers a transformative, one-time curative approach.
Mechanism of Action
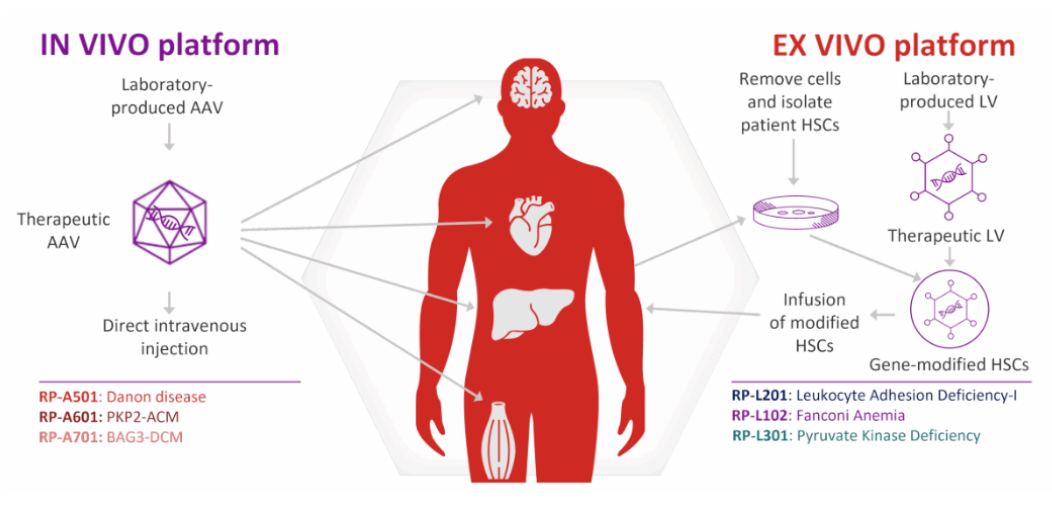
KRESLADI is a lentiviral vector (LV)-based ex vivo autologous gene therapy. It first harvests the patient’s own hematopoietic stem cells, which are then genetically modified in a highly controlled laboratory setting using a lentiviral vector to deliver a functional ITGB2 gene. The gene-corrected cells are subsequently infused back into the patient, where they reconstitute the immune system and restore the anti-infective function of leukocytes.
Clinical Data
KRESLADI demonstrated robust efficacy in a global Phase 1/2 clinical trial. Data showed that treated patients achieved a 100% overall survival rate at 12 months, with all primary and secondary endpoints successfully met. The therapy not only significantly reduced the frequency of severe infections but also greatly improved patients’ skin lesions and restored normal wound-healing capacity.
A Turbulent Regulatory Path
KRESLADI’s journey toward approval has been fraught with twists and turns, serving as a microcosm of the strict FDA oversight faced by many cutting-edge cell and gene therapies (CGT) in recent years.
Review Extension
KRESLADI’s Biologics License Application (BLA) was granted FDA Priority Review in October 2023. However, the FDA postponed the original early-2024 PDUFA date to the end of June 2024 to allow sufficient time to review additional information submitted by Rocket.
Complete Response Letter (CRL)
On June 28, 2024, the FDA issued a Complete Response Letter (CRL) to Rocket, requesting further data on chemistry, manufacturing, and controls (CMC). For gene therapies, the "product is the process" nature makes ensuring the consistency, purity, and standardized release testing of complex biological products during manufacturing often the most challenging regulatory hurdle. This setback temporarily sparked concerns in the capital markets.
Strong Comeback
Rocket did not falter. The company subsequently carried out an in-depth strategic restructuring and pipeline prioritization, focusing core resources on resolving the CMC deficiencies identified by the FDA. Following close engagement with regulators, the FDA formally accepted Rocket’s resubmission of KRESLADI’s BLA in October 2025, setting the updated PDUFA date as March 28, 2026. This key milestone largely eliminated risks related to the product’s safety or efficacy data, refocusing the primary evaluation on final manufacturing compliance.
Rocket Pharma’s R&D Platform
Rocket Pharmaceuticals’ multi-platform collaborative strategy enables it to select the most suitable delivery technology targeting the root causes of diseases: for solid organs (e.g., the heart), it uses an in vivo AAV platform for direct gene delivery; for blood and immune disorders, it employs an ex vivo LV platform to fully engineer hematopoietic stem cells. This dual-engine model forms the core competitiveness of its product pipeline, which covers a variety of complex rare diseases with high unmet clinical needs.