On March 23, Sanofi announced a global exclusive licensing agreement with California-based clinical-stage biotech firm Kali Therapeutics, obtaining the latter's next-generation trispecific T-cell engager (TCE) — KT501 — for autoimmune diseases. The deal includes a $180 million upfront and near-term payment, plus up to $1.05 billion in milestone payments, with a total value of $1.23 billion.

Transaction Details and KT501
Per the agreement terms, Sanofi secures global exclusive development and commercialization rights to KT501. In addition to the $180 million upfront payment, Kali Therapeutics is eligible for up to $1.05 billion in development and commercial milestones, as well as tiered royalties on the product's net sales in the high single-digit to low double-digit range post-launch.
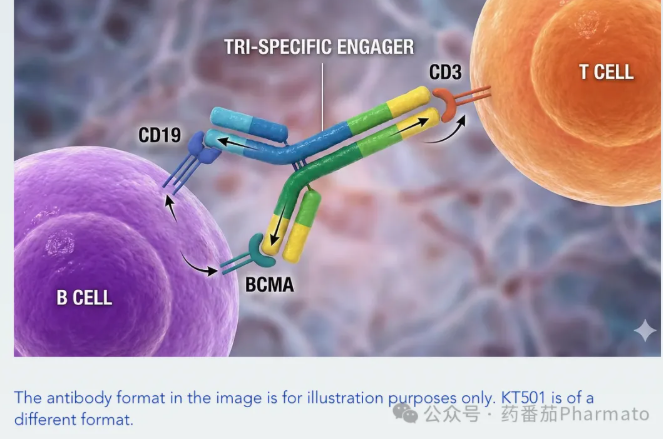
The high-value KT501 is an innovative clinical-stage IgG-like trispecific antibody. Uniquely, it binds three targets simultaneously: CD3 on T cells, and CD19 and BCMA on the B-cell lineage.
Deep "Immune Reset"
In various autoimmune diseases, pathogenic autoantibodies are driven by both B cells and differentiated plasma cells. Compared with single-target drugs, KT501 targets CD19 (covering a broad range of B-cell populations) and BCMA (targeting antibody-secreting plasma cells) concurrently, enabling more comprehensive and profound depletion of pathogenic B cells. This is expected to deliver more durable clinical responses to patients than single-target therapies.
Toxin Overcome
While TCEs exhibit potent cytotoxicity in oncology, cytokine release syndrome (CRS) induced by these agents remains a major safety concern in the autoimmune space. To address this, KT501 employs Kali's proprietary CD3 masking technology. This technique elegantly "decouples" efficacy from toxicity, maximizing cytokine reduction while ensuring robust depletion of target cells in the periphery and tissues.
Earlier this month, KT501 officially entered its First-in-Human (FIH) Phase 1a trial. The study plans to enroll approximately 24 adult rheumatoid arthritis (RA) patients with inadequate response to conventional disease-modifying antirheumatic drugs (DMARDs) to evaluate the safety, tolerability, pharmacokinetic/pharmacodynamic profiles of a single subcutaneous injection. Preliminary results are expected to be released in 2027.
Sanofi's Return to the TCE Arena
Sanofi's super "cash cow" — Dupixent, the autoimmune blockbuster with over $18 billion in annual sales — faces a patent cliff around 2030. The company is urgently seeking next-generation blockbusters to fill future revenue gaps.
For Sanofi, TCE is not an unfamiliar concept. As early as 2021, Sanofi acquired Amunix Pharmaceuticals for $1 billion to obtain its masked TCE pipeline. However, in 2024, Sanofi divested three clinical-stage oncology TCE assets (including those targeting HER2, PSMA, and EGFR) to Vir Biotechnology, temporarily strategically retreating from the TCE space.
Now, Sanofi returns to TCE with heavy investment, but this time the battlefield shifts from "oncology" to the booming "autoimmune diseases." Sanofi is committed to establishing itself as a true "powerhouse in immunology."
From late 2025 to early 2026, Sanofi has been "acquiring aggressively" in the immunology space:
- In December 2025, it spent $1.7 billion to expand collaboration with Dren Bio, focusing on myeloid cell engagers;
- Recently, it obtained global rights to the oral JAK/ROCK inhibitor rovadicitinib from China-based China Biotech at a total value of up to $1.53 billion;
- Now, it invests $1.23 billion to acquire Kali's autoimmune trispecific TCE.
About Kali Therapeutics
Kali Therapeutics, a clinical-stage biotech firm headquartered in San Mateo, California, welcomes its first large multinational pharmaceutical partner in its development history. Notably, the name "Kali" derives from the Hindu goddess of death and destruction — a fittingly robust name for an innovator dedicated to "eliminating pathogenic B cells."
Weihao Xu, CEO of Kali Therapeutics, stated in a release: "Autoimmune diseases require highly effective and safe therapeutics. KT501 effectively depletes broad B-cell populations while minimizing cytokine release, representing a major leap forward in this field. We believe Sanofi's extensive expertise in immunology and global development capabilities will accelerate the clinical progression of this promising therapy."
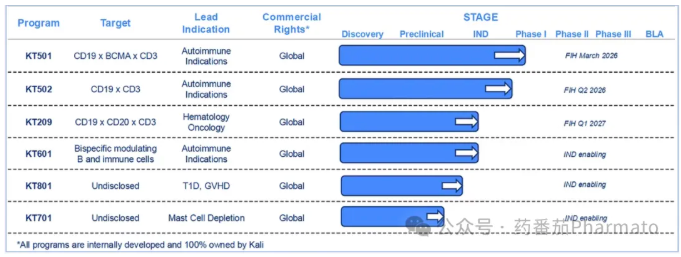
Beyond KT501, Kali's pipeline includes two other TCE products, including another trispecific antibody, and KT502, a CD19-targeting TCE slated to enter clinical development in the first half of 2026.
Conclusion
In 2026, the cross-cutting application of TCEs in autoimmune diseases has emerged as the hottest track in global pharmaceutical transactions. Giants have entered the fray, from BMS ($850 million partnership with Janux) and UCB ($110 million licensing agreement) to Boehringer Ingelheim (achieving milestone progress in collaboration with CDR-Life), and now Sanofi's $1.23 billion heavy bet.