On March 24, Shionogi announced that it will pay a $100 million upfront payment, plus $50 million in milestone payments tied to pipeline candidate SASS-002 (Sultiame) and future sales royalties, to fully acquire Apnimed’s 50% stake in Shionogi-Apnimed Sleep Science (SASS), their joint venture with the U.S. biotech firm Apnimed.
This deal, worth over $150 million in total, highlights an explosive $10-billion blue ocean in the global sleep economy: the development of oral drugs for obstructive sleep apnea (OSA).
Obstructive Sleep Apnea: The Game-Changing Potential of Oral Medications
When it comes to obstructive sleep apnea (OSA), many people first associate it with snoring. In reality, it is a serious chronic disease affecting more than 1 billion adults worldwide. Intermittent hypoxia caused by repeated collapse of upper airway muscles during sleep can lead to cardiovascular disease, hypertension, and even sudden death over the long term.
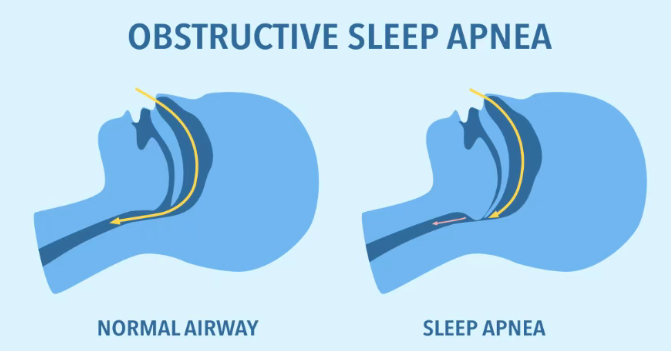
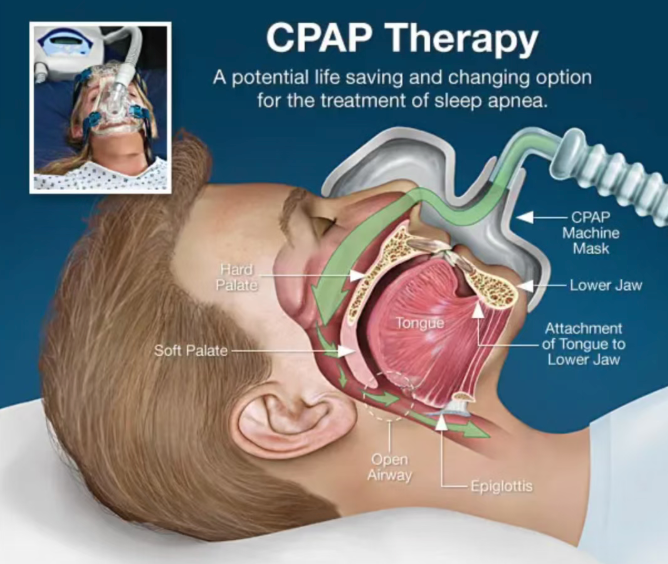
For decades, CPAP (continuous positive airway pressure) machines have been the gold-standard treatment for OSA. However, poor patient compliance—around half abandon use within one year—due to facial masks, machine noise, and poor portability has left a large unmet medical need.
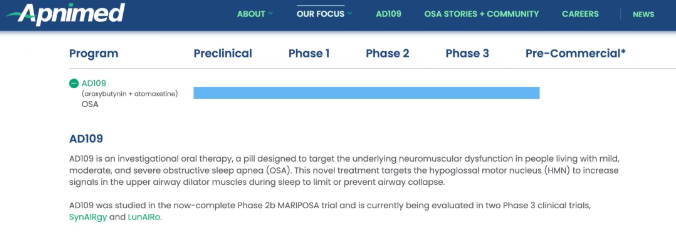
Apnimed’s lead candidate AD109, a leader in this field, is not included in this transaction.
AD109: A Neuromuscular Therapy Targeting the Root Cause
As a potential first-in-class drug, AD109 innovatively combines aroxybutynin, a novel antimuscarinic agent, with atomoxetine, a selective norepinephrine reuptake inhibitor. Unlike OSA treatments focused on weight loss, AD109 acts directly on the neuromuscular origin of upper airway collapse.
Last year, AD109 succeeded in two Phase 3 trials (SynAIRgy and LunAIRo) enrolling mild, moderate, and severe OSA patients. Apnimed plans to formally file an NDA for AD109 with the U.S. FDA in Q2 2026. If approved, it will mark an epoch-making milestone: patients could sleep soundly with just one pill before bedtime.
SASS-002 (Sultiame), acquired by Shionogi in this deal, is also highly anticipated. As a carbonic anhydrase inhibitor, it demonstrated significant potential in improving OSA in earlier Phase 2 trials and is expected to compete or complement AD109 in the future.
GLP-1 Agents Enter the Field
Notably, the OSA sector has recently welcomed a major cross-border entrant: GLP-1 weight-loss drugs. Agents such as Eli Lilly’s Zepbound (tirzepatide) greatly reduce disease severity in obese OSA patients through substantial weight loss, and received FDA approval for the OSA indication in December 2024.
Conclusion
Looking ahead, with rising public health awareness and improved diagnosis rates, market researchers project the global sleep apnea device and treatment market will exceed $10 billion after 2030. The OSA market remains in an early stage, with multiple therapeutic approaches under development.
- Diverse pathogenic mechanisms: Not all OSA patients are obese. GLP-1 agents offer little benefit for OSA caused by neuromuscular deficits or craniofacial anatomical abnormalities.
- Compliance and economics: Weight-loss injections are costly and carry gastrointestinal side effects, while small-molecule oral drugs targeting airway muscle function (such as AD109) offer unique advantages in portability, compliance, and cost.
- Combination therapy potential: For severe OSA patients, a new paradigm of “weight-loss agents as foundational therapy plus oral neuromuscular drugs for daily maintenance” is likely to emerge.