I. Background
- Urothelial carcinoma (UC) is a common type of cancer, with 614,000 new cases and 220,000 deaths from bladder cancer annually worldwide.
- 9MW2821 (Bulumtatug Fuvedotin, BFv) is a next-generation Nectin-4 targeting antibody-drug conjugate (ADC) with an MMAE payload.
- Toripalimab is a novel recombinant humanized anti-PD-1 monoclonal antibody that has been approved in China, the USA, and other countries.
- Previous studies of BFv have shown promising efficacy in patients with locally advanced or metastatic UC (la/mUC) who progressed on or after platinum-based chemotherapy and immune checkpoint inhibitors.
II. Study Design and Baseline Characteristics

- Study Design: This is a phase Ib/II, open-label, multicenter study conducted in China, consisting of dose escalation and cohort expansion phases.
- Dosing Regimen: In every 21-day cycle, patients received BFv (1.0 or 1.25 mg/kg) on days 1 and 8, and Toripalimab (240 mg) on day 1.
- Study Endpoints: The primary endpoint was safety; secondary endpoints included efficacy, pharmacokinetics (PK), and immunogenicity.
- Patient Baseline: As of April 30, 2025, 40 treatment-naïve patients with la/mUC were included in the preliminary efficacy data report. The median age of these patients was 67 years, 70.0% were male, and 82.5% were positive for Nectin-4 expression.
III. Efficacy and Safety Data
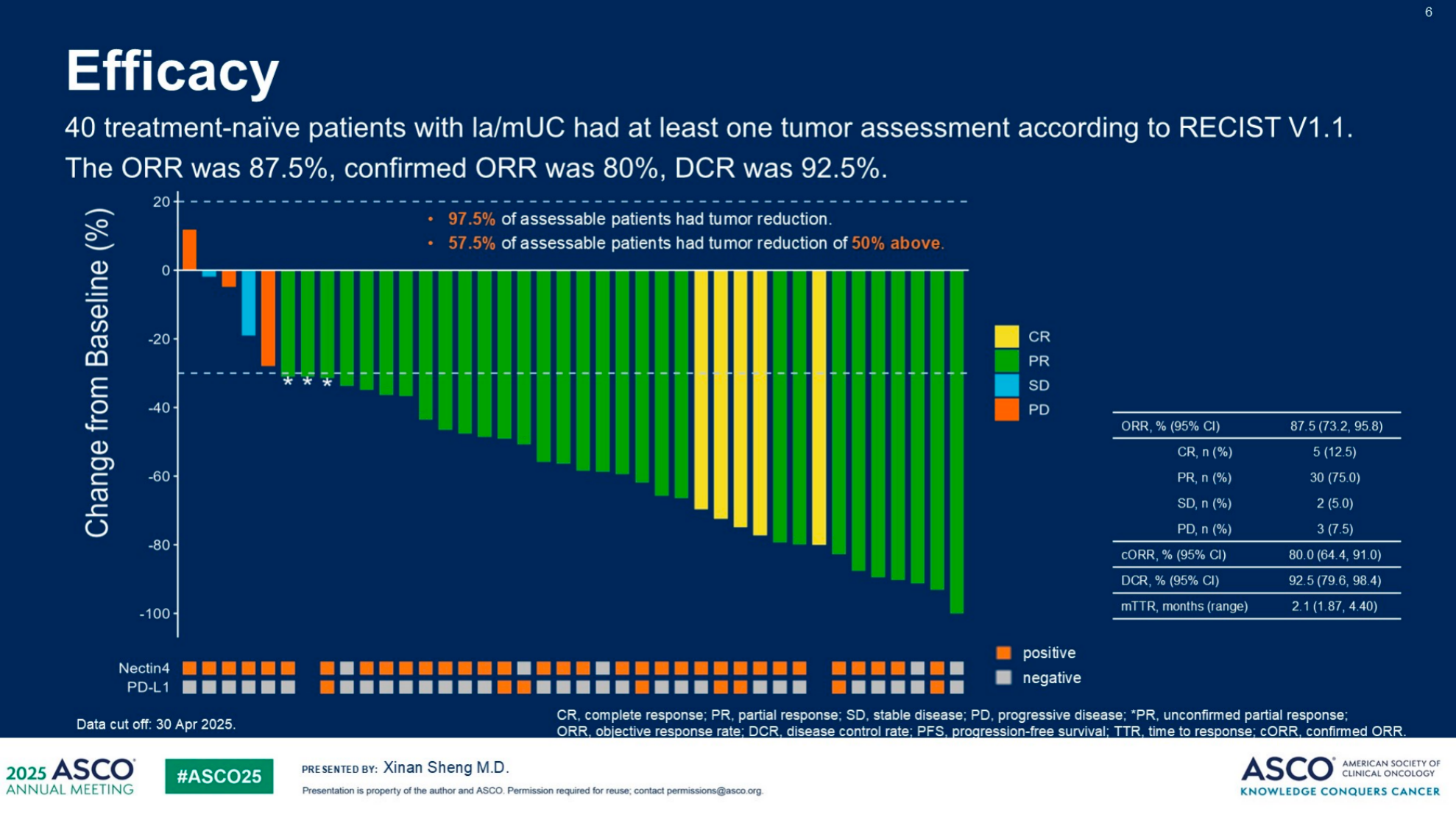
- Core Efficacy Metrics: Among the 40 treatment-naïve la/mUC patients, the objective response rate (ORR) reached 87.5%, the confirmed ORR (cORR) was 80.0%, and the disease control rate (DCR) was as high as 92.5%.
- Tumor Reduction: 97.5% of assessable patients experienced tumor reduction, with 57.5% showing a tumor reduction of 50% or more. The complete response (CR) rate was 12.5%, and the partial response (PR) rate was 75.0%.
- Subgroup Analysis: Different subgroups of treatment-naïve patients could benefit from this combination therapy regardless of primary tumor site, liver metastasis, or the expression of Nectin-4 and PD-L1, and the benefit was consistent with the overall population.
- Survival Data: With a median follow-up time of 10.8 months, the median progression-free survival (mPFS) was not yet mature, showing a preliminary result of 12.5 months (95% CI: 6.47-NA).
- Safety: The safety profile was consistent with previous trials of BFv and Toripalimab, and no new safety signals were observed in the study.
IV. Key Conclusions
The combination therapy of 9MW2821 and Toripalimab showed encouraging efficacy and a consistent safety profile in treatment-naïve patients with locally advanced or metastatic urothelial carcinoma, potentially offering a new choice for urothelial cancer patients.