On March 30, United Therapeutics announced that its inhaled formulation, Tyvaso® (treprostinil), for the treatment of idiopathic pulmonary fibrosis (IPF), successfully met the primary endpoint in the pivotal Phase 3 TETON-1 study. The results not only demonstrated superiority over placebo but also exceeded the efficacy observed in the earlier TETON-2 trial, signaling the potential to fundamentally reshape the current IPF treatment landscape.
Based on the overwhelmingly positive data from both TETON-1 and TETON-2, the company plans to submit a supplemental New Drug Application (sNDA) to the U.S. FDA before the end of summer 2026 and seek Priority Review designation, aiming to expand the approved indications of inhaled Tyvaso to include IPF as soon as possible. Currently, both the FDA and the European Medicines Agency (EMA) have granted orphan drug designation to treprostinil for the treatment of IPF.
Clinical Data: Significant Improvement in Lung Function, Exceeding Expectations
TETON-1 is a 52-week, multicenter, randomized, double-blind, placebo-controlled Phase 3 registrational study that enrolled 598 IPF patients across North America (the United States and Canada). The primary objective was to evaluate the safety and efficacy of inhaled Tyvaso.
- Primary endpoint achieved:
Compared with placebo, patients treated with Tyvaso showed a statistically significant improvement of 130.1 mL in absolute forced vital capacity (FVC) at Week 52 (p < 0.0001), indicating a substantial slowing of the rapid decline in lung function. - Reduced risk of disease progression:
Tyvaso demonstrated a statistically significant reduction in the risk of clinical worsening. - Secondary endpoints all positive:
Tyvaso showed numerical improvements over placebo across multiple key secondary endpoints, including:- Time to first acute exacerbation of IPF
- Change in percent predicted FVC
- K-BILD (King’s Brief Interstitial Lung Disease Questionnaire) score
- Diffusing capacity for carbon monoxide (DLCO)
- Time to first acute exacerbation of IPF
- Change in percent predicted FVC
- K-BILD (King’s Brief Interstitial Lung Disease Questionnaire) score
- Diffusing capacity for carbon monoxide (DLCO)
- Time to first acute exacerbation of IPF
- Change in percent predicted FVC
- K-BILD (King’s Brief Interstitial Lung Disease Questionnaire) score
- Diffusing capacity for carbon monoxide (DLCO)
In addition, treatment benefits were consistent across all patient subgroups. Regardless of background antifibrotic therapy (such as nintedanib or pirfenidone), smoking status, or the need for supplemental oxygen, Tyvaso demonstrated stable efficacy. The drug was also well tolerated, with no new safety signals identified.
Integrated Analysis of TETON-1 and TETON-2
Previously, the TETON-2 study conducted outside North America had already achieved significant success, with full results recently published in the New England Journal of Medicine (NEJM).
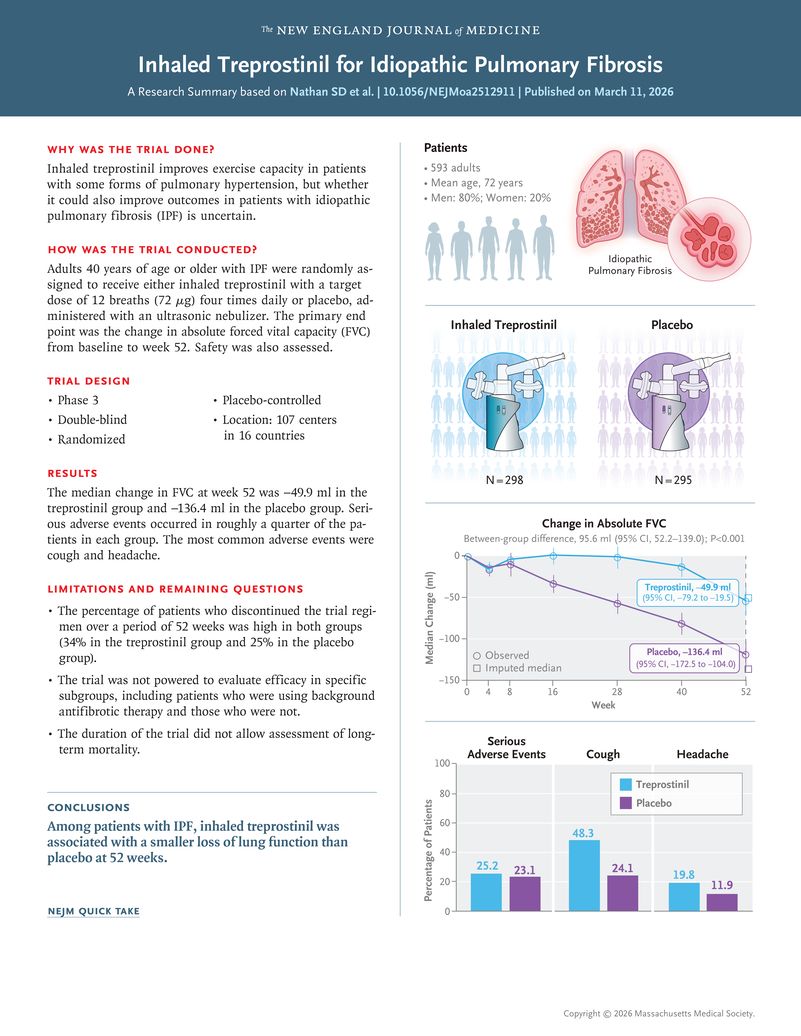
The pooled analysis of TETON-1 and TETON-2 further reinforces the robustness of Tyvaso:
- Combined FVC improvement:
The treatment effect on absolute FVC change at Week 52 reached 111.8 mL (p < 0.0001). - Favorable survival trend:
Although overall survival at Week 52 did not reach statistical significance, the trend clearly favored the Tyvaso treatment group.
These findings suggest that Tyvaso delivers consistent efficacy across geographies and diverse IPF patient populations, standing up to large-scale clinical validation.
Why Tyvaso Could Be a “Game Changer”
Currently available IPF therapies—primarily oral antifibrotic agents—are often associated with significant gastrointestinal side effects and poor patient adherence. Tyvaso’s breakthrough performance can be attributed to its unique delivery method and multi-mechanistic action:
- Direct delivery to the lungs:
The inhaled formulation allows the drug to act directly at the site of disease (the lungs), increasing local drug concentration while minimizing systemic side effects. - Multi-pathway mechanism:
According to Dr. Peter Smith, Senior Vice President of Product Development, inhaled Tyvaso not only delivers drug directly to the lungs but also targets fibrosis, vascular remodeling, and inflammation simultaneously—addressing key pathological pathways that existing IPF therapies fail to fully cover.
With its strong efficacy, favorable safety profile, and differentiated mechanism, Tyvaso is well positioned to become a transformative therapy in the treatment of idiopathic pulmonary fibrosis.